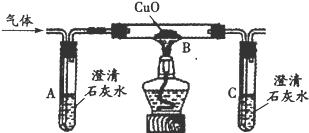
��Ŀ����
ij������ȤС���H2O2�ķֽ�������������ʵ��̽����
��1���±��Ǹ�С���о�Ӱ��������⣨H2O2���ֽ����ʵ�����ʱ�ɼ���һ�����ݣ�
��10mL H2O2��ȡ150mL O2�����ʱ�䣨�룩
�ٸ��о�С������Ʒ���ʱ��������Ũ�ȡ� �� �����ضԹ�������ֽ����ʵ�Ӱ�죮
�ڴ�����Ӱ���������ֽ����ʵ���������ѡһ����˵�������ضԷֽ������к�Ӱ�죿 ��
��2����������ͬ���ۼ�״̬��ͬ��MnO2�ֱ���뵽5mL 5%��˫��ˮ�У����ô����ǵ�ľ
�����ԣ��ⶨ������£�
��д��H2O2�����Ļ�ѧ��Ӧ����ʽ ��
��ʵ����˵���������õĴ�С�� �йأ�
��1���±��Ǹ�С���о�Ӱ��������⣨H2O2���ֽ����ʵ�����ʱ�ɼ���һ�����ݣ�
��10mL H2O2��ȡ150mL O2�����ʱ�䣨�룩
| Ũ�� ʱ�䣨�룩 ��Ӧ���� |
30%H2O2 | 15%H2O2 | 10%H2O2 | 5%H2O2 |
| ������������ | ��������Ӧ | ��������Ӧ | ��������Ӧ | ��������Ӧ |
| ���������� | 360 | 480 | 540 | 720 |
| MnO2���������� | 10 | 25 | 60 | 120 |
�ڴ�����Ӱ���������ֽ����ʵ���������ѡһ����˵�������ضԷֽ������к�Ӱ�죿
��2����������ͬ���ۼ�״̬��ͬ��MnO2�ֱ���뵽5mL 5%��˫��ˮ�У����ô����ǵ�ľ
�����ԣ��ⶨ������£�
| ������MnO2�� | ������� | �۲��� | ��Ӧ��������ʱ�� |
| ��ĩ״ | ��ϲ��� | ���ҷ�Ӧ�������ǵ�ľ����ȼ | 3.5���� |
| ��״ | ��Ӧ���������Ǻ�����ľ��δ��ȼ | 30���� |
��ʵ����˵���������õĴ�С��
��������1������������Һ��ʵ������ȡ��������ҪҩƷ���ڳ����º��ѷֽ�õ���������ֽ��ٶ���Ũ�ȡ��¶ȡ����������ص�Ӱ�����ʵ�鷽����֤��ʱ��Ҫע��ʵ��Ŀ��Ʊ�������ȷ��ʵ������ȷ�ԣ�
��2�����������ǹ�������ֽ�Ĵ������ɴ�����ľ����ȼ����֪����ɴ˿�д������ʽ����ʵ�������֪���������ô�С��Ӱ�����أ�
��2�����������ǹ�������ֽ�Ĵ������ɴ�����ľ����ȼ����֪����ɴ˿�д������ʽ����ʵ�������֪���������ô�С��Ӱ�����أ�
����⣺��1���ٸ��ݱ��и��������ݣ����������ȵ�����£���ͬŨ�ȵĹ���������Һ���Ǽ�������Ӧ�����������ȵ�����£���ͬŨ�ȵĹ���������Һ���ֽ⣬˵����������ķֽ��������¶��йأ����ǵõ���ͬ�����ʱ�䲻ͬ��Ũ��Խ��Ӧ���ٶ�Խ�죬˵����������ķֽ�������Ũ���йأ��Ƚ�ͬһŨ�ȵĹ���������Һ��30%ʱ�����������ȵ�ʱ����Ҫʱ����360s���д������ȵ������£���Ҫʱ����10s��˵����������ķֽ��������¶ȡ������йأ��ʴ�Ϊ���¶ȣ�������
�ڷ����������ݿ��Կ�����Ũ��Խ��Ӧ����Խ�죬�����ܼӿ��������ķֽ⣬�д����Ƿֽ����ʿ죬
�ʴ�Ϊ���¶�����ѧ��Ӧ���ʼӿ죻��Ӧ��Ũ������ѧ��Ӧ���ʼӿ죻ʹ�ú��ʵĴ�����ѧ��Ӧ���ʼӿ죻
��2����H2O2�ڶ������������·�����Ӧ�Ļ�ѧ��Ӧ����ʽΪ��2H2O2
2H2O+O2�����ʴ�Ϊ��2H2O2
2H2O+O2����
����������������ͬʱ����ĩ״�������̱ȿ�״�������̷�Ӧ����ʱ��̣�˵���Ӵ�����Է�Ӧ������Ӱ�죬�ʴ�Ϊ���Ӵ������
�ڷ����������ݿ��Կ�����Ũ��Խ��Ӧ����Խ�죬�����ܼӿ��������ķֽ⣬�д����Ƿֽ����ʿ죬
�ʴ�Ϊ���¶�����ѧ��Ӧ���ʼӿ죻��Ӧ��Ũ������ѧ��Ӧ���ʼӿ죻ʹ�ú��ʵĴ�����ѧ��Ӧ���ʼӿ죻
��2����H2O2�ڶ������������·�����Ӧ�Ļ�ѧ��Ӧ����ʽΪ��2H2O2
| ||
| ||
����������������ͬʱ����ĩ״�������̱ȿ�״�������̷�Ӧ����ʱ��̣�˵���Ӵ�����Է�Ӧ������Ӱ�죬�ʴ�Ϊ���Ӵ������
����������ͨ��ͼ�����ݣ���������������Է�Ӧ���ʵ�Ӱ�죬�Ѷ����У�Ҫע���������������ͬ��ֻ��һ�������ı�ʱ�����Ӱ�췴Ӧ���ʣ�

��ϰ��ϵ�д�
 Сѧѧϰ�ð���ϵ�д�
Сѧѧϰ�ð���ϵ�д� Сѧͬ�����������ܾ�ϵ�д�
Сѧͬ�����������ܾ�ϵ�д�
�����Ŀ
��ѧ������Ȥ�С���ͬѧ��ij����������������������ɷ�����ȡ������Ʒ��������ϡ�������ձ��з�Ӧ�����������е����ʲ����ᷴӦ��Ҳ������ˮ������ʵ���������±������йط�Ӧ�Ļ�ѧ����ʽΪ��Fe+2HCl=FeCl2+H2����
| ��Ӧǰ | ��Ӧ�� | ||
| ʵ������ | ϡ��������� | ������Ʒ������ | ��Ӧ���ձ��л��������� |
| 200g | 14.5g | 214.0g | |
��2�������������Ʒ����������������
�����⣺
���ʵ����У�������ȤС���ij������������ʯ��Ʒ��������ʵ�顢���������㣬�˽��˸ø���������ԭ�ϡ���Ʒ�������Ϣ����������������ǵ�ʵ���������ش�
ȡ�ó���ij������ĩ28.36g������ֻ��Fe��C�������������г�ַ�Ӧ��������������ͨ�������ij���ʯ��ˮ�У��õ�3g��ɫ������
��1������˸�����ĩ������̼������֮�ȣ�
��2����ȡ���ݲ�ͬ�����ĸ�����ĩ�ֱ�ӵ�50g����������ͬ�������У���ַ�Ӧ��õ�ʵ���������±���ʾ������״���£�2g H2�����Ϊ22.4 L��
����ݱ������ݼ���������H2SO4������������
��3������ʵ����м�������m g������ĩ�����㷴Ӧ������ʣ��Ĺ�������Ϊ���٣����ú�m�Ĵ���ʽ��ʾ��
���ʵ����У�������ȤС���ij������������ʯ��Ʒ��������ʵ�顢���������㣬�˽��˸ø���������ԭ�ϡ���Ʒ�������Ϣ����������������ǵ�ʵ���������ش�
ȡ�ó���ij������ĩ28.36g������ֻ��Fe��C�������������г�ַ�Ӧ��������������ͨ�������ij���ʯ��ˮ�У��õ�3g��ɫ������
��1������˸�����ĩ������̼������֮�ȣ�
��2����ȡ���ݲ�ͬ�����ĸ�����ĩ�ֱ�ӵ�50g����������ͬ�������У���ַ�Ӧ��õ�ʵ���������±���ʾ������״���£�2g H2�����Ϊ22.4 L��
| ʵ����� | �� | �� | �� |
| ���������ĩ������/g | 2.836 | 5.672 | 8.508 |
| ����������������״����/L | 1.12 | 2.24 | 2.80 |
��3������ʵ����м�������m g������ĩ�����㷴Ӧ������ʣ��Ĺ�������Ϊ���٣����ú�m�Ĵ���ʽ��ʾ��