��Ŀ����
��֪A��BΪ���ֺ�ɫ��ĩ��DΪ��ɫ������C��F������ɫ���壬A��B��C��D��E��F��������֮���ת����ϵ����ͼ��ʾ����������������ʡ�ԣ�
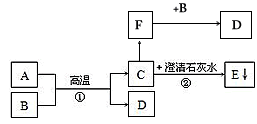
��ش�
��1��B�Ļ�ѧʽΪ___________��D�Ļ�ѧʽΪ___________��
��2����Ӧ�ٵĻ�����Ӧ������____________�� ��Ӧ�ڵĻ�ѧ����ʽΪ________________________________�� ��������Ӧ��A��F������___________�ԡ�
��3������ֻ����ҽѧ�о���С����ֱ����ʢ��C��F������ƿ�У�š��ƿ�ǣ������ֻС����ܿ춼���ˡ����С������������Ҫԭ��__________�����ͬ����ͬ������
��1��B�Ļ�ѧʽΪ___________��D�Ļ�ѧʽΪ___________��
��2����Ӧ�ٵĻ�����Ӧ������____________�� ��Ӧ�ڵĻ�ѧ����ʽΪ________________________________�� ��������Ӧ��A��F������___________�ԡ�
��3������ֻ����ҽѧ�о���С����ֱ����ʢ��C��F������ƿ�У�š��ƿ�ǣ������ֻС����ܿ춼���ˡ����С������������Ҫԭ��__________�����ͬ����ͬ������
��1��CuO��Cu
��2���û���Ӧ��CO2+Ca(OH)2==CaCO3��+H2O����ԭ
��3����ͬ
��2���û���Ӧ��CO2+Ca(OH)2==CaCO3��+H2O����ԭ
��3����ͬ

��ϰ��ϵ�д�
�����Ŀ

 ��֪A��BΪ���ֺ�ɫ��ĩ��DΪ��ɫ���ʣ�A��B��C��D��E��������֮���ת����ϵ����ͼ��ʾ��
��֪A��BΪ���ֺ�ɫ��ĩ��DΪ��ɫ���ʣ�A��B��C��D��E��������֮���ת����ϵ����ͼ��ʾ��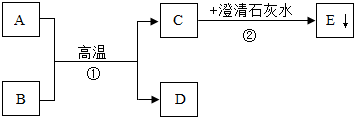
 ��֪A��BΪ���ֺ�ɫ��ĩ��DΪ��ɫ���ʣ�A��B��C��D��E��F��������֮���ת����ϵ��ͼ��ʾ����ش�
��֪A��BΪ���ֺ�ɫ��ĩ��DΪ��ɫ���ʣ�A��B��C��D��E��F��������֮���ת����ϵ��ͼ��ʾ����ش� ��֪A��BΪ���ֺ�ɫ��ĩ��DΪ��ɫ���ʣ�A��B��C��D��E��������֮���ת����ϵ��ͼ��ʾ����ش�
��֪A��BΪ���ֺ�ɫ��ĩ��DΪ��ɫ���ʣ�A��B��C��D��E��������֮���ת����ϵ��ͼ��ʾ����ش�