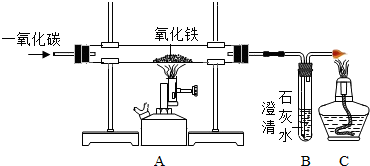
��Ŀ����
 ����H2SO4��CuSO4�Ļ����Һ��Ϊ�˷��������Һ��H2SO4��CuSO4�������������������ʵ�鷽����ȡ�ķݻ����Һ��100g���ֱ������뵽50g��100g��150g��200gijNaOH��Һ�У��������ʵ�����������
����H2SO4��CuSO4�Ļ����Һ��Ϊ�˷��������Һ��H2SO4��CuSO4�������������������ʵ�鷽����ȡ�ķݻ����Һ��100g���ֱ������뵽50g��100g��150g��200gijNaOH��Һ�У��������ʵ�����������| �ڢ��� | �ڢ��� | �ڢ��� | �ڢ��� | |
| NaOH��Һ������/g | 50 | 100 | 150 | 200 |
| ���ɳ���������/g | 0 | 2.45 | 7.35 | 9.8 |
��2������������Һ��CuSO4���������������Ƕ��٣���д��������̣�
��3�������Һ��H2SO4��������������Ϊ
��4������100g�����Һ�в��ϼ�������NaOH����Һ���뻭������NaOH��Һ�����������������������Ĺ�ϵͼ�����ڴ��������ͼ����ͼ��
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ���,�й��������������ļ���
ר�⣺�ۺϼ��㣨ͼ���͡������͡��龰�ͼ����⣩
�����������������ͭ�Ļ����Һ�μ�����������Һ�����ᡢ����ͭ�������������Ʒ�����Ӧ����������Ĵ�����������������ͭ��������������ͭ�����������ᷴӦ����ܲ���������ͭ���������Լ�¼�����У�����50.0g����������Һʱ��������������Ϊ0�����ڼ�������������Һ200.0g�Ժ�����������ٱ仯��˵������ͭҲ����ȫ��Ӧ�������ɳ��������ֵΪ9.8g��
��������ͭ���������Ʒ�Ӧ�Ļ�ѧ����ʽ���ɳ���������ͭ�������ɼ�������Һ������ͭ��������������ͭ������Ӧ���������Ƶ�������
�����������������Ʒ�Ӧ�Ļ�ѧ����ʽ���ɻ����Һ�����������������֮������Ӧ���������Ƶ�������
���������ἰ����ͭ��Ӧ���������Ƶ�������������������Һ���������������ıȿɼ�����������������Һ��������
��������ͭ���������Ʒ�Ӧ�Ļ�ѧ����ʽ���ɳ���������ͭ�������ɼ�������Һ������ͭ��������������ͭ������Ӧ���������Ƶ�������
�����������������Ʒ�Ӧ�Ļ�ѧ����ʽ���ɻ����Һ�����������������֮������Ӧ���������Ƶ�������
���������ἰ����ͭ��Ӧ���������Ƶ�������������������Һ���������������ıȿɼ�����������������Һ��������
����⣺��1���ɼ�¼���ݱ���֪��������ͭ��ȫ��Ӧ�����ɳ���9.8g������9.8g��
��2�������Һ��CuSO4������Ϊx������������������Ϊy��
CuSO4+2NaOH�TCu��OH��2��+Na2SO4
160 80 98
x y 9.8g
=
��
=
x=16g y=8g��
�����Һ��CuSO4���������������ǣ�
��100%=16%��
�𣺻����Һ��CuSO4����������������16%��
�ɵڢ���͵ڢ���ʵ�����ݿ�֪50g����������Һ�����ɳ���������7.35g-2.45g=4.9g��������9.8g�����õ�����������Һ��������100g��
NaOH��Һ��������������Ϊ��
��100%=8%
��3�������Һ��H2SO4������Ϊz���ɵڢ��顢�ڢ���͵ڢ���ʵ�����ݿ�֪�������ĵ�����������Һ����Ϊ75g��
H2SO4+2NaOH�TNa2SO4+2H2O
98 80
z 75g��8%
=
z=7.35g
�����Һ��H2SO4��������������Ϊ��
��100%=7.35%��
�𣺻����Һ��H2SO4��������������Ϊ7.35%��
��4��ץס��㡢�۵㼰�������ƣ�������㣺���ᱻ��ȫ��Ӧ����������������ͭ��Ӧ������ɫ������ͭ��������ʱ����������������Һ������75g��
�����۵㣺��Һ������ͭ��ȫ��Ӧ���ټ�������������Һ����������ͭ�������ӣ���ʱ����������������Һ������100g+75g=175g��
�������ƣ�����175g����������Һ������ͭ��ȫ��Ӧ������ټ��������������Һ���ٷ�����Ӧ������������ͭ�������䣻
�ʴ�Ϊ��
��2�������Һ��CuSO4������Ϊx������������������Ϊy��
CuSO4+2NaOH�TCu��OH��2��+Na2SO4
160 80 98
x y 9.8g
| 160 |
| x |
| 98 |
| 9.8g |
| 80 |
| y |
| 98 |
| 9.8g |
x=16g y=8g��
�����Һ��CuSO4���������������ǣ�
| 16g |
| 100g |
�𣺻����Һ��CuSO4����������������16%��
�ɵڢ���͵ڢ���ʵ�����ݿ�֪50g����������Һ�����ɳ���������7.35g-2.45g=4.9g��������9.8g�����õ�����������Һ��������100g��
NaOH��Һ��������������Ϊ��
| 8g |
| 100g |
��3�������Һ��H2SO4������Ϊz���ɵڢ��顢�ڢ���͵ڢ���ʵ�����ݿ�֪�������ĵ�����������Һ����Ϊ75g��
H2SO4+2NaOH�TNa2SO4+2H2O
98 80
z 75g��8%
| 98 |
| z |
| 80 |
| 75g��8% |
z=7.35g
�����Һ��H2SO4��������������Ϊ��
| 7.35g |
| 100g |
�𣺻����Һ��H2SO4��������������Ϊ7.35%��
��4��ץס��㡢�۵㼰�������ƣ�������㣺���ᱻ��ȫ��Ӧ����������������ͭ��Ӧ������ɫ������ͭ��������ʱ����������������Һ������75g��
�����۵㣺��Һ������ͭ��ȫ��Ӧ���ټ�������������Һ����������ͭ�������ӣ���ʱ����������������Һ������100g+75g=175g��
�������ƣ�����175g����������Һ������ͭ��ȫ��Ӧ������ټ��������������Һ���ٷ�����Ӧ������������ͭ�������䣻
�ʴ�Ϊ��

�������ڽ���漰��Ӧ����������ʱ��Ҫע��������㡢�۵���������壬���Ϊ��Ӧ��ʼ�������۵����Ǹ÷�Ӧǡ����ȫ��Ӧ��

��ϰ��ϵ�д�
�����Ŀ
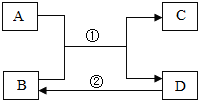 A��B��C��D�dz��л�ѧ�������ʣ�����֮�������·�Ӧ��ת����ϵ����Ӧ����δ�������
A��B��C��D�dz��л�ѧ�������ʣ�����֮�������·�Ӧ��ת����ϵ����Ӧ����δ�������