��Ŀ����
ijͬѧΪ�˲ⶨNaC l��MgCl2����������MgCl2��������������������ʵ�飺��80g������������ˮ�����Һ��ƽ���ֳ��ķݣ��ֱ������ͬ����������NaOH��Һ���������ʵ�����ݣ�
l��MgCl2����������MgCl2��������������������ʵ�飺��80g������������ˮ�����Һ��ƽ���ֳ��ķݣ��ֱ������ͬ����������NaOH��Һ���������ʵ�����ݣ�
| ʵ����� | һ | �� | �� | �� |
| ���������������g�� | 20 | 20 | 20 | 20 |
| ����NaOH��Һ��������g�� | 20 | 40 | 60 | 80 |
| ���ɳ�����������g�� | 2.9 | m | 8.7 | 8.7 |
�ʣ�
����m��ֵΪ ��
ԭ����������MgCl2�����������Ƕ��٣���Ҫ��д��������̣�
���𰸡���1��5.8������2��71.25%
��������
�����������1���ӱ������ݿ�֪����һ�����ɳ���Ϊ2.9g���ڶ��μ���������������Һ�������ǵ�һ�ε�2���������ɳ���������Ҳ�ǵ�һ�ε�2����ӦΪ2.9g��2=5.8g��
��2���Ƚ����������ε����ݿ�֪�������η�Ӧ�Ѿ���ȫ��������֪���������ɳ���������Ϊ8.7g��������MgCl2��Mg(OH)2��������ϵ������⡣�������������£�
�⣺��ԭ����������MgCl2������Ϊx
MgCl2 + 2NaOH==Mg(OH)2�� + 2NaCl
95 58
x 8.7g
95:58=x:8.7g
x=14.25g
ԭ����������MgCl2����������Ϊ��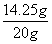 ��100%=71.25%
��100%=71.25%
��ԭ����������MgCl2����������Ϊ71.25%

 ��ѧ����ͬ����ϰϵ�д�
��ѧ����ͬ����ϰϵ�д�����Ҫ�ɷ֣���̼���ơ������ᣩ���ڳ�����ˮ����״�ǵĵײ���������ˮ��Ѹ��ð�����ݡ�Ϊ�˽��䷴Ӧ���̣�����ѧ ϰС���ͬѧ����������̽����
ϰС���ͬѧ����������̽����

���̽��һ����ʦ�ṩ��һƿ��̼���ƣ�Na2CO4����С��ͬѧ����������ʵ��̽����
���������1����ɫ��ҺX�е���������Щ���ʣ�
����������衿��������ʵ��������ɫ��ҺX��һ�����ڵ������� ��
���������غ㶨�ɣ�ͬѧ�Ƕ���ҺX�л����ܴ����������ʣ��������²��룺
��Na2CO3 ����NaHCO3 ����Na2CO3 �� NaHCO3�Ļ���
���������ϡ�
| NaHCO3��Һ | Na2CO3��Һ | |
| ����ϡ���� | ����� | �������� |
| ���뱥��ʯ�� | ��Һ����� | ����� |
| ����CaCl2��Һ | ���������� | ��Һ����� |
| ������Һ�����ڣ� ������ͨ�����ʯ��ˮ | ����ʯ��ˮ����� | ����ʯ��ˮ�����Ա仯 |
��ʵ��̽����Ϊ��ȷ����ҺX�����ܴ��ڵ��������ʣ�������·���������һ����ɡ�
| ���� | ���� | �����뷽��ʽ |
| ��ȡ��������ˮ���������CaCl2��Һ�� | ������ɫ���� | �÷�Ӧ����ʽ�� |
| �ڽ�������Ӧ����Һ���ˣ�ȡ��Һ �� |
| ֤������۳����� |
����˼��ʵ�鲽����У��Ȼ�����Һ������Ŀ���� ��
���̽������
�������2����β���Na2CO3 �� NaHCO3�Ĺ��������и��ɷֵ�������
��1��Ϊ����������и��ɷֵ�������С�������ͼ1װ�á�ͨ������Bװ�õ������仯��ȷ����Ʒ�и��ɷֵ���������װ�����������ã�ϡ��������Ʒ������װ��B��CO2��NaOH����ȫ��Ӧ������������η��֣��ⶨ�����ƫ����ƫ���ԭ���У�
�� ���� ��

��2������ʦ��ָ����С�����������ͼ2��ʵ��װ�ý���ʵ�顣С��ȡ9.5g������Ʒ��
��ʵ�飬ʵ����Ϻ���Bװ������4.4g������Ʒ�и��ɷֵ������ֱ�Ϊ���٣���д����
����̣�����������һλС������
�������õ�����Է���������Na2CO3��106��NaHCO3��84��CO2��44��

 �IJ�������Һ�в��ϼ�����������
�IJ�������Һ�в��ϼ����������� ��
��  D.��̼�����������������仯
D.��̼�����������������仯

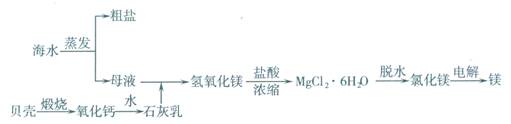 ��ˮ���ۺ����ÿ����Ʊ�����þ������������ͼ��ʾ��
��ˮ���ۺ����ÿ����Ʊ�����þ������������ͼ��ʾ��