��Ŀ����
4����ѧ����������ϢϢ��أ������û�ѧ֪ʶ�ش��������⣮��1��þȼ�պ����ɵĹ�������Ϊʲô��ԭ���������������Ϊ���ɹ�����������ڲμӷ�Ӧ�Ĺ���þ��μӷ�Ӧ�������������ͣ�
��2���ճ���ˮһ�������Ե�ԭ����CO2+H2O=H2CO3���û�ѧ����ʽ��ʾ����
��3���ô����ǵ�ľ����������C+O2 $\frac{\underline{\;��ȼ\;}}{\;}$CO2���û�ѧ����ʽ��ʾ����
��4���ҹ����������������ʪ��ұ����ȡͭ�Ĺ��ң���������ͭ��Һ��Ӧ�Ļ�ѧ����ʽ��Fe+CuSO4�TFeSO4+Cu��
��5�������ȼ�յ�ʵ��ʱΪʲôҪ�ڼ���ƿ����װһЩˮSO2+H2O=H2SO3���û�ѧ����ʽ��ʾ����
���� ��1����ѧ��Ӧ��ѭ�����غ㶨�ɣ����μӷ�Ӧ�����ʵ�����֮�ͣ����ڷ�Ӧ�����ɵ����ʵ�����֮�ͣ�
��2�����ݶ�����̼��ˮ������̼����з�����
��3������̼�Ļ�ѧ���ʷ������
��4����������ͭ��Һ��Ӧ��������������Һ��ͭ��д����Ӧ�Ļ�ѧ����ʽ���ɣ�
��5�����ݶ�����������ʷ�����
��� �⣺��1��þ���ڿ�����ȼ��ʱ��þ�Ϳ����е�������Ӧ��������þ�����ɵ�����þ���������ڲμӷ�Ӧ��þ������������������֮�ͣ�����������������ԭ��þ������������
��2��������̼����ˮ��Ӧ����̼�ᣬ̼����������ԣ�
��3��ľ������Ҫ�ɷ�Ϊ̼����̼��������ȼ�����������ɶ�����̼��
��4����������ͭ��Һ��Ӧ��������������Һ��ͭ����Ӧ�Ļ�ѧ����ʽΪ��Fe+CuSO4�TFeSO4+Cu��
��5����Ϊ��ȼ�����ɶ��������������ж�������ˮ��Ӧ���������
�𰸣���1����Ϊ���ɹ�����������ڲμӷ�Ӧ�Ĺ���þ��μӷ�Ӧ�������������ͣ�
��2��CO2+H2O=H2CO3��
��3��C+O2 $\frac{\underline{\;��ȼ\;}}{\;}$CO2��
��4��Fe+CuSO4=FeSO4+Cu��
��5��SO2+H2O=H2SO3��
���� �����ѶȲ����������÷��ӵĻ������ʷ����ͽ������ķ�������ѧ����ʽ����д�������������ֵĴ����в����Ͽ���ʵ�������������غ㶨�ɡ���д������������ŵȣ��ǽ�������Ĺؼ���

| A�� |  | B�� |  | C�� |  | D�� |  |
| A�� |  ��ȡʳ�� | B�� |  ϡ��Ũ���� | C�� |  �ⶨ��Һ��pH | D�� |  ����ҩƷ��ȡ�� |
| A�� | �μӷ�Ӧ��O2��������11.2g | B�� | ��������̼����Ԫ����������1��3 | ||
| C�� | ����CO2��H2O�ķ��Ӹ�����Ϊ2��3 | D�� | �������в�����Ԫ�� |
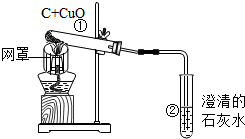 ��ľ̿��ԭ����ͭ��ʵ����ͼ��
��ľ̿��ԭ����ͭ��ʵ����ͼ��