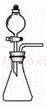��Ŀ����
��֪̼��������270�����Ҿ��ֽܷ�Ϊ̼���ơ�ˮ�Ͷ�����̼����̼�������Ȳ��ֽ⣮����ij������������һ��̼�������л���������̼���ƣ�Ϊ�˲ⶨ��Ʒ��̼��������������������ļ��鲽�����£�
��ȡһֻ�ྻ����������������Ϊag���������м�����Ʒ���Ƶ�������Ϊm1g��
�ڼ��ȸ�ʢ����Ʒ��������
�۽����������ȴ������������ʣ������������
�ܶ���ظ�����ں͢������أ��Ƶ�������ʣ������������Ϊm2g��
��1��д��̼���������ȷֽ�Ļ�ѧ����ʽ����
��2�������������������ƣ������Ⱥ�������ŵ������У�����ţ���ȴ��
��3���������⣬��a��m1��m2�Ĵ���ʽ��ʾ��Ʒ��̼�����Ƶ���������Ϊ������
��4������١��ۺܶ͢���Ҫ�õ�����Ϊ0.1g��������ƽ�������������У�m1﹣m2����ֵ����0.6g��������Ʒ��̼�����Ƶ���������Ϊ90%�������������Ʒ���ٿˣ�
�⣺��1��̼��������270�����Ҿ��ֽܷ�Ϊ̼���ơ�ˮ�Ͷ�����̼����Ӧ�Ļ�ѧ����ʽΪ2NaHCO3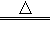 Na2CO3+H2O+CO2����
Na2CO3+H2O+CO2����
��2��ʹ������ǯ�Ѽ��Ⱥ���������������C�н�����ȴ����ֹ��ȴ���������տ����е�ˮ�֣���Ӱ�����ij����Ľ����
��3������Ʒ��̼�����Ƶ�����Ϊx
2NaHCO3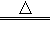 Na2CO3+H2O+CO2�� ������������
Na2CO3+H2O+CO2�� ������������
168 106 168﹣106=62
x m1﹣m2
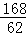 =
=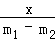
x=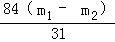
��Ʒ��̼�����Ƶ���������=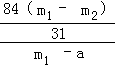 ��100%=
��100%=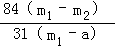
�ʴ�Ϊ��
��1��2NaHCO3 Na2CO3+H2O+CO2����
Na2CO3+H2O+CO2����
��2������ǯ��C��
��3��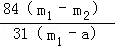 ��
��
��4��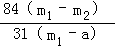 ��100%=90%
��100%=90%
��m1﹣a��=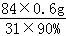 ��1.8g
��1.8g
�����������Ʒ1.8g��

�����йز��������ܵ�ʹ��ͼʾ��ȷ���ǣ�ʡ�Լгֺͼ���װ�ã���������
| �� | A�� |
����ˮ������ | B�� |
�����ſ��������� |
| �� | C�� |
�ø������������ | D�� |
���������̼ |
����ʳƷ���ζƷ�����������У�û�����������ɵ���( )
A��ţ�̱����� B����ˮ��ե�ɹ�֭
C����ʳ��� D������ۺ�С�մ�����ͷ
�����и�����������У�����Ϊ����ȷ���ǣ�������
| �� | A�� | ���ɱ������ʷ�����ͬ����ѧ���ʲ�ͬ |
| �� | B�� | ������ռ���ʷ�����ͬ����ѧ������ͬ |
| �� | C�� | ���ʯ��ʯī���������ʲ�ͬ�����Ԫ����ͬ��ԭ�����з�ʽ��ͬ |
| �� | D�� | CO��CO2����ѧ���ʲ�ͬ�����Ԫ����ͬ�����ӽṹ��ͬ |


 B��
B��
 D��
D��