��Ŀ����
�±���һЩ���ӵİ뾶����λ��10-10 m��
���������ݿ��Կ�����
��1���˵������ͬ�ĵ�ԭ�Ӻ����ӵĴ�С˳��Ĺ�������ʲô��
��2����ͬ�������Ҹ�������Ų���ͬ�����ӣ��뾶��С˳��Ĺ�������ʲô��
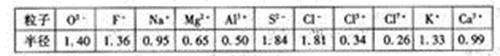
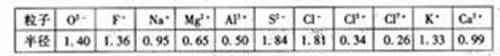
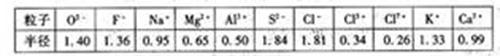
| �� | O2- | F- | Na+ | M2+ | Al3+ | S2- | Cl- | Cl5+ | Cl7+ | K+ | Ca2+ |
| �뾶 | 140 | 1.36 | 0.95 | 0.65 | 0.50 | 1.84 | 1.81 | 0.34 | 0.26 | 1.33 | 0.99 |
��1���˵������ͬ�ĵ�ԭ�Ӻ����ӵĴ�С˳��Ĺ�������ʲô��
��2����ͬ�������Ҹ�������Ų���ͬ�����ӣ��뾶��С˳��Ĺ�������ʲô��
�����������������еĵ�����������������������ͼ���е���������ɸ���Ľ��
����⣺��1���Ƚ�ͼ��Cl-��Cl5+��Cl7+���������ݣ�����֪�����ǵİ뾶�����μ�С�ģ����Կ����жϺ˵������ͬ�ĵ�ԭ�����ӣ�������Խ�࣬�뾶Խ��
��2��O2-��F-��Na+��Mg2+��Al3+�����ǵĺ������������˵�����������ӣ����Ǻ����������ȣ���������������п��Կ��������ǵİ뾶�����μ�С�ģ��ʿ��Եó�����������Ų���ͬ�����ӣ��˵����Խ�뾶ԽС��
�ʴ�Ϊ����1���˵������ͬ�ĵ�ԭ�����ӣ��˵������ͬ�ĵ�ԭ�����ӣ�������Խ�࣬�뾶Խ��
��2����������Ų���ͬ�����ӣ��˵����Խ�뾶ԽС��
��2��O2-��F-��Na+��Mg2+��Al3+�����ǵĺ������������˵�����������ӣ����Ǻ����������ȣ���������������п��Կ��������ǵİ뾶�����μ�С�ģ��ʿ��Եó�����������Ų���ͬ�����ӣ��˵����Խ�뾶ԽС��
�ʴ�Ϊ����1���˵������ͬ�ĵ�ԭ�����ӣ��˵������ͬ�ĵ�ԭ�����ӣ�������Խ�࣬�뾶Խ��
��2����������Ų���ͬ�����ӣ��˵����Խ�뾶ԽС��
������֪������ԭ�ӵĻ�������ͬʱ��ס����֮��Ĺ�ϵ��ԭ������=������=�������������ȷԭ�Ӻ�����֮��Ĺ�ϵ�����ܹ�����������������ó����ɣ�

��ϰ��ϵ�д�
 �Ƹ�С״Ԫ�������������ϵ�д�
�Ƹ�С״Ԫ�������������ϵ�д� ����һ������ܼƻ�ϵ�д�
����һ������ܼƻ�ϵ�д�
�����Ŀ