��Ŀ����
16����1����֪��ˮ���ܶ�Ϊ1.0g/mL��ʵ��������50g������������Ϊ5%���Ȼ�����Һ����Ҫ��ȡ�Ȼ���2.5g����Ҫ��ȡˮ47.5mL����2�����ƵIJ���ʾ��ͼ���£���ʵ�����ȷ����˳��ΪCBDEA������ĸ��ţ���
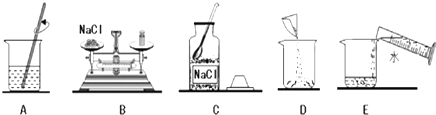
��3������⣬��������Һ��������������ƫС����ԭ������Ǣ٢ڢۣ�����ţ���
���Ȼ��ƹ��岻�� �ڳ���ʱ�����������������ͬ��ֽƬ
����ȡˮʱ�����Ӷ��� ��װƿʱ����������Һ����
��4����ҵ�����г���10%��ϡ������������Ʒ��������⣬ʵ������Ҫ���Ƹ�����200g����������Ͳ��ȡ������������Ϊ38%��Ũ���ᣨ�ܶ�Ϊ1.19g/mL��44.2mL��������������0.1����
���� �������е���Һ���Ƶ�֪ʶ���з�������ܼ�������=��Һ����-����������������Һ���ƵIJ�����������������ƫС���������ʼ�С�����ܼ������Ե�ʣ�ϡ����Һǰ�������������䣬�ݴ˽��
��� �⣺��1��ˮ������Ϊ��50g-2.5g=47.5g����ˮ�����Ϊ��$\frac{47.5g}{1g/mL}$=47.5mL�����47.5mL��
��2��������Һʱ�����ȴ��Լ�ƿ��ȡʳ�ι��壬Ȼ�������ƽ�ϳ������ٷŽ��ձ��У�Ȼ����ȡ�õ�ˮ�����ձ��У�����ò����������ܽ⣬���CBDEA��
��3�����Ȼ��ƹ��岻��������ʳ�ε�����ƫС����������������С��
�ڳ���ʱ�����������������ͬ��ֽƬ����ҩƷ��ʳ�ε���������ֽƬ��������������˵�������ʳ�ε�������С����������������С��
����ȡˮʱ�����Ӷ���������ˮ�࣬��������������С��
��װƿʱ����������Һ��������Ӱ����������������
����٢ڢۣ�
��4������ҪŨ��������Ϊx
x��1.19g/mL��38%=200g��10%
x=44.4mL
���44.2��
���� ���⿼�������Һ�����ƣ���ɴ��⣬�����������е���Һ���ƵIJ����Լ������������������ı�����ؽ��У�

 ��У����ϵ�д�
��У����ϵ�д�| A�� | ��Һ������ɫ������Һ�� | B�� | ��������Һ����ϡ��Һ | ||
| C�� | ��Һ�в���ͬʱ���������� | D�� | ��Һ�и�����������ͬ |
| A�� | 20mLˮ�� 20mL�ƾ���Ϻ����С��40mL������Ϊ������Ŀ���� | |
| B�� | �����㲻���������˵���˷����ڲ����˶� | |
| C�� | ʯī��һ��������ת��Ϊ���ʯ�ǻ�ѧ�仯������Ϊ̼ԭ�ӵ����з�ʽ�����˸ı� | |
| D�� | ˮֱͨ����ֽ⣬����Ϊˮ���ӱ��ƻ����γ��µķ��� |

| A�� | ��Ӧǰ��ԭ������������ı� | |
| B�� | �÷�Ӧ���û���Ӧ | |
| C�� | ���ɵ�C��D������Ϊ14��27 | |
| D�� | �μӷ�Ӧ���������ʷ��Ӹ�����Ϊ4��1 |
��1�������±����ṩ����Ϣ����д�йغ�̼���ʵĶ�Ӧ���ʣ�
| ������; | ʯī���缫 | ���ʯ�и�� | ����̿��ˮ |
| ��Ӧ���� | ������ | Ӳ�ȴ� | ������ |
A��Һ̬������̼��������Ⱦ��������
B��������̼�ɸ�����ȼ������棬��������
C��Һ̬������̼����ʱ���ȣ������˿�ȼ����Ż��
��3����Ȼ������Ҫ�ɷ��Ǽ��飨CH4����д�������ڿ�������ȫȼ�յĻ�ѧ����ʽΪCH4+2O2$\frac{\underline{\;��ȼ\;}}{\;}$CO2+2H2O�������ŷŶ�����̼����������ЧӦ��������ǻ���Ѱ�Ҳ���̼Ԫ�ص�ȼ�ϣ��ռ���������Դ����̫���ܡ�ˮ�ܡ��ȣ���̼�����ϸ�������ҵ�һ����̼�ٴ���ֲ�����֣�
