��Ŀ����
���Ż�ѧ��ҵ�ķ�չ����ǰ��չ��CH4��CO2��Ϊԭ�ϵġ�C1��ѧ����Ϊ���������ı�Ȼ���ƣ���ν��C1��ѧ�����Է�����ֻ��һ��̼ԭ�ӵĻ������CO2��CH4�ȣ�Ϊԭ�����ϳ�һϵ�л���ԭ�Ϻ�ȼ�ϵĻ�ѧ��ͨ����Ȼ���к���H2S���ж����壬��ͼ1������Ȼ��Ϊԭ�����ϳ�����ȼ�϶����ѣ�CH3OCH3���Ĺ������̣�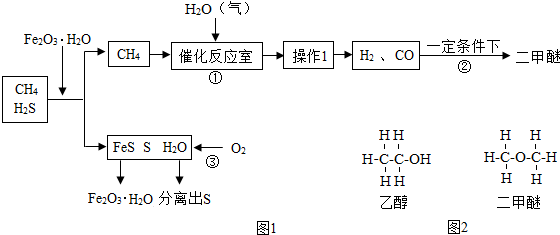
��1�����������Ҵ��Ļ�ѧʽ��ͬ������C2H6O�����ӽṹ��ͼ2�����ǵķ��ӽṹ ��ѡ���ͬ������ͬ������
��2����д����Ӧ�ڵĻ�ѧ����ʽ��
��3���Ӵ���Ӧ�ҳ����������к���������CO2��H2O���������ʲ���1��Ҫ��Ϊ�˳��ӣ�����Ϊ����1�ľ�������ǣ� ��
��4��ʹ��Fe2O3?H2O��ȥH2S���������к���Ԫ�ص������� �֣������в���ѭ���������� ���ѧʽ����

��1�����������Ҵ��Ļ�ѧʽ��ͬ������C2H6O�����ӽṹ��ͼ2�����ǵķ��ӽṹ
��2����д����Ӧ�ڵĻ�ѧ����ʽ��
��3���Ӵ���Ӧ�ҳ����������к���������CO2��H2O���������ʲ���1��Ҫ��Ϊ�˳��ӣ�����Ϊ����1�ľ�������ǣ�
��4��ʹ��Fe2O3?H2O��ȥH2S���������к���Ԫ�ص�������
���㣺���ʵ��ת�����Ʊ�,��������ļ�������ӷ���,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺���ʵ��Ʊ�
��������1���Ƚ϶�������ƾ��Ļ�ѧʽ��ԭ�ӵ����༰������
��2����֪��Ӧ��������д���÷�Ӧ�Ļ�ѧ����ʽ��2CO+4H2
CH3OCH3+H2O
��3�����dz���H2O��������
��4��ʹ��Fe2O3?H2O��ȥH2S�����������غ㶨�ɣ��Ʋ��������к���Ԫ�ص����ʺ������в���ѭ�������ʣ�
��2����֪��Ӧ��������д���÷�Ӧ�Ļ�ѧ����ʽ��2CO+4H2
| ||
��3�����dz���H2O��������
��4��ʹ��Fe2O3?H2O��ȥH2S�����������غ㶨�ɣ��Ʋ��������к���Ԫ�ص����ʺ������в���ѭ�������ʣ�
����⣺
��1���ɶ�������ƾ��Ļ�ѧʽ��ԭ�ӵ����༰������ͬ��֪����������ƾ����Ҵ�C2H5OH���Ļ�ѧʽ�����ͬ��
��2��һ����̼�������ڴ����������¿����ɶ����ѣ�ͬʱ����һ�ֿɲ������ѭ����������ΪҺ̬�������д���÷�Ӧ�Ļ�ѧ����ʽ2CO+4H2
CH3OCH3+H2O��
��3���Ӵ���Ӧ�ҳ����������к���������CO2��H2O���������ʲ���1��Ҫ��Ϊ�˳��ӣ�����Ϊ����1�ľ�������Ǹ��
��4��ʹ��Fe2O3?H2O��ȥH2S���������к���Ԫ�ص�����Ϊ��������ϡ���ᣬ����2�֣������в���ѭ���������� Fe2O3?H2O��
�ʴ�Ϊ��
��1����ͬ����2��2CO+4H2
CH3OCH3+H2O����3�������4��2��Fe2O3?H2O��
��1���ɶ�������ƾ��Ļ�ѧʽ��ԭ�ӵ����༰������ͬ��֪����������ƾ����Ҵ�C2H5OH���Ļ�ѧʽ�����ͬ��
��2��һ����̼�������ڴ����������¿����ɶ����ѣ�ͬʱ����һ�ֿɲ������ѭ����������ΪҺ̬�������д���÷�Ӧ�Ļ�ѧ����ʽ2CO+4H2
| ||
��3���Ӵ���Ӧ�ҳ����������к���������CO2��H2O���������ʲ���1��Ҫ��Ϊ�˳��ӣ�����Ϊ����1�ľ�������Ǹ��
��4��ʹ��Fe2O3?H2O��ȥH2S���������к���Ԫ�ص�����Ϊ��������ϡ���ᣬ����2�֣������в���ѭ���������� Fe2O3?H2O��
�ʴ�Ϊ��
��1����ͬ����2��2CO+4H2
| ||
�������������Ĺؼ��Ǹ�������ṩ����Ϣ���ҳ���Ӧ������д����Ӧ�ķ���ʽ���ٸ��ݷ���ʽ�����й�����ķ������жϣ�

��ϰ��ϵ�д�
�����Ŀ
���л����У����ڸ��Ϸ��ϵ��ǣ�������
| A��Ca3��PO4��2 |
| B��CO��NH2��2 |
| C��K2SO4 |
| D��KNO3 |
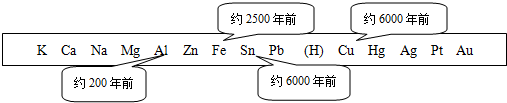
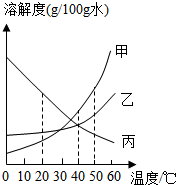 �ܽ�ȿɱ�ʾ�����ܽ��ԵĴ�С��
�ܽ�ȿɱ�ʾ�����ܽ��ԵĴ�С��