��Ŀ����
��̼������һ���Ե��ܺĺ�Ч�ܵ�Ϊ��Ҫ�������Խ��ٵ����������ŷŻ�ýϴ�������¾��÷�չģʽ�����ʣ�
��1�������������ж�����̼����������������Ҫ�Ƕ�����̼��Ȼ�������Ķ�����̼��;����Ҫ�� ��
��2����ѧ�������о���������̼����ת���ɼ��飨CH4�����״���CH3OH�������ᣨCH3COOH���Ȼ���ԭ�ϣ���Щ����ԭ������ �������������л����������
��3�����������в����ϡ���̼���á�������ǣ�����ţ� ��
�ٸ������̭���ܺġ�����Ⱦ��ҵ���ڴ�����չ�������磻�����ƺͿ�������Դ�����ͳ��Դ�����Ż�������ƣ���ǿ������Ȼ�ɹ⣬���������õ磮
��1�������������ж�����̼����������������Ҫ�Ƕ�����̼��Ȼ�������Ķ�����̼��;����Ҫ��
��2����ѧ�������о���������̼����ת���ɼ��飨CH4�����״���CH3OH�������ᣨCH3COOH���Ȼ���ԭ�ϣ���Щ����ԭ������
��3�����������в����ϡ���̼���á�������ǣ�����ţ�
�ٸ������̭���ܺġ�����Ⱦ��ҵ���ڴ�����չ�������磻�����ƺͿ�������Դ�����ͳ��Դ�����Ż�������ƣ���ǿ������Ȼ�ɹ⣬���������õ磮
���㣺��Ȼ���е�̼ѭ��,�л��������������
ר�⣺̼�����뺬̼���������������;
��������1�����ݽ����������ж�����̼����������������Ҫԭ���ǻ�ʯȼ�ϵ�ȼ�ա���Ȼ�������Ķ�����̼����Ҫ;����ֲ��Ĺ�����ý��н��
��2�����ݼ��飨CH4�����״���CH3OH�������ᣨHCOOH���Ȼ���ԭ�϶�����̼Ԫ�ؽ��н��
��3�����ݡ���̼���á�����Ҫ���ٶ�����̼���ŷŽ��н��
��2�����ݼ��飨CH4�����״���CH3OH�������ᣨHCOOH���Ȼ���ԭ�϶�����̼Ԫ�ؽ��н��
��3�����ݡ���̼���á�����Ҫ���ٶ�����̼���ŷŽ��н��
����⣺
��1�������������ж�����̼����������������Ҫԭ���ǻ�ʯȼ�ϵ�ȼ�գ���Ȼ�������Ķ�����̼����Ҫ;����ֲ��Ĺ�����ã�
��2�����飨CH4�����״���CH3OH�������ᣨHCOOH���Ȼ���ԭ�϶�����̼Ԫ�أ����������л������
��3������̼���á�����Ҫ���ٶ�����̼���ŷţ����Ը������̭���ܺġ�����Ⱦ��ҵ�����ƺͿ�������Դ�����ͳ��Դ���Ż�������ƣ���ǿ������Ȼ�ɹ⣬���������õ綼�ܼ��ٶ�����̼���ŷţ����ϡ���̼���á������������չ�������粻����ٶ�����̼���ŷţ������ϡ���̼���á����
�ʴ�Ϊ��
��1��ֲ��Ĺ�����ã�
��2��������
��3���ۣ�
��1�������������ж�����̼����������������Ҫԭ���ǻ�ʯȼ�ϵ�ȼ�գ���Ȼ�������Ķ�����̼����Ҫ;����ֲ��Ĺ�����ã�
��2�����飨CH4�����״���CH3OH�������ᣨHCOOH���Ȼ���ԭ�϶�����̼Ԫ�أ����������л������
��3������̼���á�����Ҫ���ٶ�����̼���ŷţ����Ը������̭���ܺġ�����Ⱦ��ҵ�����ƺͿ�������Դ�����ͳ��Դ���Ż�������ƣ���ǿ������Ȼ�ɹ⣬���������õ綼�ܼ��ٶ�����̼���ŷţ����ϡ���̼���á������������չ�������粻����ٶ�����̼���ŷţ������ϡ���̼���á����
�ʴ�Ϊ��
��1��ֲ��Ĺ�����ã�
��2��������
��3���ۣ�
��������̼�����ǵ�����ȵ㻰�⣬ͬѧ��Ҫ���յ�̼��Ҫ���ǽ��ܼ��ţ�

��ϰ��ϵ�д�
�����Ŀ
�Ŵ��������鶾����ԭ����4Ag+2H2S+O2�T2X+2H2O��X�Ļ�ѧʽΪ��������
| A��Ag2O |
| B��AgS |
| C��Ag2O2 |
| D��Ag2S |
������O3�����������ȣ�ClO2�����������⣨H2O2�����������е������������������ж����У�������
| A�������� | B�������� |
| C������ | D����Ԫ�� |
��ͼΪС��������ԭ��ģ�ͣ���Ȧ��С��Ϊ���ӣ���ȦΪԭ�Ӻˣ�˵����ȷ���ǣ�������
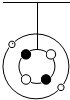

| A����ģ�ͱ�ʾһ��̼ԭ�� |
| B����ԭ�ӵĺ˵����Ϊ4 |
| C����ԭ��������Ҫ������2�������� |
| D����ԭ�Ӻ���2�����Ӻ�2�����ӹ��� |
���ڿ����ijɷֲ���ȷ���ǣ�������
| A������ 78% |
| B������31% |
| C��ϡ������0.94% |
| D��������̼0.03% |
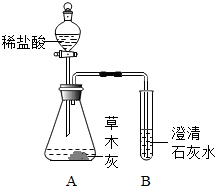 С��ͬѧ�ڳԷ�ʱ��С�Ľ����������ڻ�¯�ԵIJ�ľ���ϣ������д������ݲ�������һ���������Բ�ľ�ҳɷֽ���̽������Ȥ��
С��ͬѧ�ڳԷ�ʱ��С�Ľ����������ڻ�¯�ԵIJ�ľ���ϣ������д������ݲ�������һ���������Բ�ľ�ҳɷֽ���̽������Ȥ��