��Ŀ����
19����ͼ��ʾΪʵ�����г����������Ʊ����������ռ�������ʵ��IJ����������Ը�����ĿҪ�ش��������⣺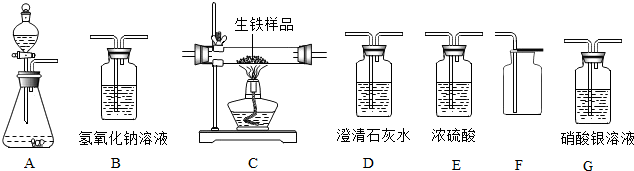
��1������ʯ��ʯ��ϡ����Ϊԭ�ϣ���ʵ�������Ʊ����ռ�һƿ���﴿���Ķ�����̼���壮
����ѡ����������˳��ΪAGEF ����д���������ĸ����
����д��װ��A����������Ӧ�Ļ�ѧ����ʽCaCO3+2HC1=CaCl2+H2O+CO2����
�ۼ��鼯��ƿ�ж�����̼�������ռ����ķ�������һ��ȼ�ŵ�ľ���ŵ�����ƿ�ڣ�ľ������Ϩ��˵��������̼�����Ѽ�����
��2�������ֶ���������Ҫ�Ͻ�ij��ѧ�о�С���ͬѧҪ֤��ij������Ʒ�к���̼Ԫ�أ����ⶨ�京̼��������������˳��Ϊ��A��E��C��D1��B��D2����������A�е�ҩƷΪ˫��ˮ�Ͷ������̣���ȡ10g����Ʒ�ڴ�����ȼ�գ��������ĿҪ�����й����ݣ��ش��������⣺
����д��װ��A����������Ӧ�Ļ�ѧ����ʽ2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2����
������װ��E������������ˮ������
�۵�����D1�г��ֳ���ʯ��ˮ����ǵ��������֤��������Ʒ�к���̼Ԫ�أ�
�ܵ�����װ���ڵĻ�ѧ��Ӧ����ȫ���к���������D1����m1g������B����m2g������D2����û�б仯����10g�������к�̼Ԫ�ص�����Ϊ$\frac{3��m_{1}+m_{2}��}{11}$g����������Ϊ������ʽ����
���� ��1��ʵ����ͨ���ô���ʯ��ʯ��ʯ��ϡ���ᷴӦ��ȡ������̼����Ӧ����Ҫ���ȣ�����ʯ��ʯ��ʯ����Ҫ�ɷ���̼��ƣ��ܺ�ϡ���ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼��
������̼����ȼ�գ���֧��ȼ�գ�
��2��ͨ������£����������ڶ������̵Ĵ������£��ֽ�����ˮ��������
Ũ���������ˮ�ԣ�������������������������������̼�����壻
������̼��ʹ����ʯ��ˮ����ǣ�
����װ�������仯��������Լ���������к�̼Ԫ�ص�������
��� �⣺��1������ѡ����������˳��Ϊ��ͨ��Aװ����ȡ�Ķ�����̼����Gװ���г�ȥ�Ȼ������壬��ͨ��Eװ�ó�ȥˮ���������ͨ��Fװ�����������ſ������ռ�������Ķ�����̼���壻
��װ��A����������Ӧ�Ļ�ѧ����ʽΪ��CaCO3+2HC1=CaCl2+H2O+CO2����
�ۼ��鼯��ƿ�ж�����̼�������ռ����ķ����ǣ���һ��ȼ�ŵ�ľ���ŵ�����ƿ�ڣ�ľ������Ϩ��˵��������̼�����Ѽ�����
���AGEF��CaCO3+2HC1=CaCl2+H2O+CO2������һ��ȼ�ŵ�ľ���ŵ�����ƿ�ڣ�ľ������Ϩ��˵��������̼�����Ѽ�����
��2����װ��A����������Ӧ�Ļ�ѧ����ʽΪ��2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2����
������װ��E������������ˮ������
�۵�����D1�г��ֳ���ʯ��ˮ����ǵ��������֤��������Ʒ�к���̼Ԫ�أ�
�ܾ���������D1����m1g������B����m2g������D2����û�б仯��˵�����ɶ�����̼������Ϊ��m1g+m2g��
��10g�������к�̼Ԫ�ص�����Ϊ����m1g+m2g����$\frac{12}{44}$��100%=$\frac{3��m_{1}+m_{2}��}{11}$��
���2H2O2$\frac{\underline{\;MnO_2\;}}{\;}$2H2O+O2��������ˮ����������ʯ��ˮ����ǣ�$\frac{3��m_{1}+m_{2}��}{11}$��
���� ʵ������������֮������õ����ڱ��֣����Ҫѧ�����ʵ�顢�۲�ʵ�顢����ʵ�飬Ϊ��ʾ����֮������õ�ʵ�ʵ춨������

| A�� | ���õ����Ż���ˮ���� | |
| B�� | �ƾ����������Ż�������ʪ������ | |
| C�� | Ũ����մ��Ƥ���ϣ������ô�����ˮ��ϴ | |
| D�� | ͼ�����ͼ���Ż���Һ̬������̼��������� |
| A�� | ��ʳ�ú����ʳ�������Ч��ֹ��״���״� | |
| B�� | ���̵������ж����������̿��Ե������ж����� | |
| C�� | ���������dz�����Σ�Ӧ�ʵ���������㡢����Ⱥ������ʶ��ʳ�� | |
| D�� | ��ά�ء����۶��������࣬������̼���⡢��Ԫ�� |

| A�� | �����Ӻ�����18������ | B�� | ���������ڷǽ���Ԫ�� | ||
| C�� | �������Ǵ������������ | D�� | �����Ӻ������������Ӳ� |
| A�� | 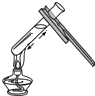 ��ʢ��Һ����Թܼ��� | B�� | 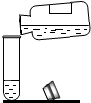 ���Թ����㵹Һ��ҩƷ | ||
| C�� |  ���װ�õ������� | D�� | 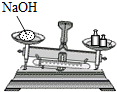 �����������ƹ��� |
| A�� | ���嶡���������л��� | |
| B�� | һ�����嶡���ѷ��Ӻ���5��̼ԭ�� | |
| C�� | һ�����嶡���ѷ�������ԭ�Ӻ���ԭ�Ӹ�����12��1 | |
| D�� | ���嶡��������5��̼ԭ�ӡ�12����ԭ�Ӻ�1����ԭ�ӹ��ɵ� |
 ��ijЩ��ѧ֪ʶ��ͼʽ��ʾ���ȼ����ֶ�Ҫ����ͼ��һ������ת��������ͼʽ����������ʾ��Ӧһ�����ת����
��ijЩ��ѧ֪ʶ��ͼʽ��ʾ���ȼ����ֶ�Ҫ����ͼ��һ������ת��������ͼʽ����������ʾ��Ӧһ�����ת����


