��Ŀ����
2��ˮ������֮Դ����ش��������⣮��1��ͼ1Ϊ���ˮ��װ�ã����ˮ�Ĺ����й۲쵽�������ǣ���
�Ⱥ�˳����д�����������������ݲ��������Թ���Һ���½�����a��������������Լb�������������Ķ�����
�Թ�b���ռ��������������������ˮ�Ļ�ѧ����ʽ��2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2����
��2��ˮ����������ˮ�ľ���������ͼ2��ʾ��
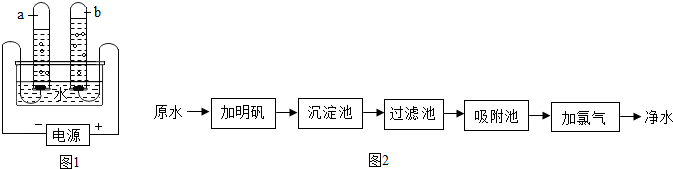
������ˮ����������ˮʱ��ʹ�õľ�ˮ������ABE��
A������ B������ C����� D������ E������
�����������м������̿����д�������ƣ���������ȥˮ�е���ζ����������������������������仯����ѧ�仯������
������ˮ�к�������Ca��HCO3��2�ȿ����Ի������ˮʱCa��HCO3��2�����ֽⷴӦ�����������Ե�̼��ơ�ˮ�Ͷ�����̼������Ǻ��г���ˮ����ԭ��֮һ����д����ˮʱCa��HCO3��2���ȷֽ�Ļ�ѧ����ʽCa��HCO3��2$\frac{\underline{\;\;��\;\;}}{\;}$CaCO3��+H2O+CO2����
���� ��1�������ڵ��ˮʵ���У��ɹ۲쵽�������У���������������٣���ʹ�����ǵ�ľ����ȼ����������������࣬��ȼ�գ��������������ٵĶ�������������������Ƴ������������������������������������������������ܽ�һ���Ƴ�ˮ����Ԫ�غ���Ԫ����ɽ��з�����
��2���ٸ��ݳ��������ˡ������ȷ�����������������ˮ���з�����
�ڸ��ݻ���̿�����������ý��
�۸���̼������ڼ��ȵ����������������Ե�̼��ơ�ˮ�Ͷ�����̼���з�����
��� �⣺��1�������ˮʵ�顱�۲쵽�������������������ݲ������Թ���Һ���½���a������������Լ��b�������������Ķ�����
��������������٣���ʹ�����ǵ�ľ����ȼ������֪��b������Ϊ���������������ɽ������ǵ�ľ�������Թ��У�ľ����ȼ��˵��Ϊ��������������������࣬��ȼ�գ�����a������Ϊ������ˮ��ͨ��������������������������ʷ�Ӧ�Ļ�ѧ����ʽΪ��2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2����
��2������ͼ�п�֪������ˮ����������ˮʱ��ʹ�õľ�ˮ�����г��������ˡ���������ѡ��ABE��
�ڻ���̿�����������ã��ܹ�����ɫ�غ���ζ��û�����������ɣ����������仯��
��̼������ڼ��ȵ����������������Ե�̼��ơ�ˮ�Ͷ�����̼����ѧ����ʽΪ��Ca��HCO3��2$\frac{\underline{\;\;��\;\;}}{\;}$CaCO3��+H2O+CO2����
�ʴ�Ϊ����1���������������ݲ��������Թ���Һ���½�����a��������������Լb�������������Ķ�����������2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2����
��2����ABE��
�ڻ���̿��������
��Ca��HCO3��2$\frac{\underline{\;\;��\;\;}}{\;}$CaCO3��+H2O+CO2����
���� �����Ҫ���վ���ˮ�ķ����ȷ����֪ʶ��ֻ���������ܶ���ط��������������ȷ���жϣ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | ʵ�� | B�� | ���� | C�� | �۲� | D�� | ���� |
| A�� | 20% | B�� | 16.7% | C�� | 83.3% | D�� | 25% |
| A�� | ������ͥ�������������ռ� | |
| B�� | �Ͻ����սոѣ����Ϳ�����PM2.5��Ũ�� | |
| C�� | ��չ��̼���ã���С��������վռ�ܷ������ı��� | |
| D�� | �û�����δ����ŷŵķ�ˮ���ũ�� |
| A�� | �������� | B�� | ʪ�·�ɹ�� | C�� | ��ʯ���� | D�� | ���������ڻ� |
��ѧ���� | 2N | $\stackrel{+2}{Mg}$ | 3H2 | 2H2O |
��ʾ���� | 2����ԭ�� | +2�۵�þԪ�� | 3������� | 2��ˮ���� |
