��Ŀ����
2�����������DZ���������Դ��һ����Ч;����ij��ҵ���Ϻ�����ͭ��������������������ɵõ�ͭ������������Һ����Ҫ�������£�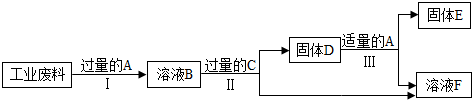
��֪��CuO+H2SO4�TCuSO4+H2O Fe+Fe2��SO4��3�T3FeSO4
��1�����̢���������������Ӧ�Ļ�ѧ����ʽ��Fe2O3+3H2SO4�TFe2��SO4��3+3H2O��
��2�����̢��ַ�Ӧ��Ӧ�е�ʵ������ǹ��ˣ�
��3��A��F�У�������ͭ����Ԫ�ص���BD������ĸ��ţ���
���� ��1�����������������ᷴӦ������������ˮ���з�����
��2�����ݷ��벻���Թ����Һ��ʹ�ù��˵ķ������з�����
��3�����ݷ����ƶϣ�A�к������ᣬB�к�������ͭ����������C������D�к�������ͭ��E��ͭ��F�к��������������з�����
��� �⣺��1��Ҫ���յõ��������������ܽ�����ͭ��������ʹ�õ�����ϡ���ᣬ����������ϡ���ᷴӦ������������ˮ����ѧ����ʽΪ��Fe2O3+3H2SO4=Fe2��SO4��3+3H2O��
��2�����̢��ַ�Ӧ��Ҫ�õ��������Һ����Ҫ�������˲�����
��3�����ݷ����ƶϣ�A�к������ᣬB�к�������ͭ����������C������D�к�������ͭ��E��ͭ��F�к���������������ѡ��BD��
�ʴ�Ϊ����1��Fe2O3+3H2SO4�TFe2��SO4��3+3H2O��
��2�����ˣ�
��3��BD��
���� ���⿼���˽��������ʣ��ѶȲ�����ɴ��⣬�����������е�֪ʶ���У�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
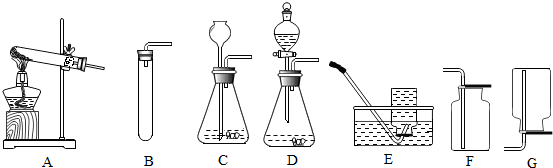
5������ʵ����������������ǣ�������
| A�� | ��ѧʵ��̽����ɺ�ʣ��ҩƷӦ����ָ�������� | |
| B�� | �ý�ͷ�ιܵμ�Һ��ʱ���ι�Ӧ���������������Ϸ� | |
| C�� | ���Թ����Һ�����ʱ��Һ������ﵽ�Թ��ݻ��Ķ���֮һ | |
| D�� | ϡ��Ũ����ʱ����Ũ�������ձ�������ע��ˮ�в����Ͻ��� |
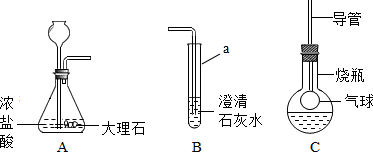
6����ͼ��ʾװ���ж�����;������˵��������ǣ�������

| A�� | ϴ������ȥCO�е�HCl��װ����ӦʢH2O | |
| B�� | ���飺֤��CO�л���CO2��װ����Ӧʢ����ʯ��ˮ | |
| C�� | �����������ſ������ռ��ȿ����������ʱ������Ӧ��װ�õ�A��ͨ�� | |
| D�� | ��Ҫ�ⶨʵ�����Ƶõ�����02���������Ӧ��ƿ����װ��ˮ���ٽ�O2��B��ͨ�� |
10������˵����ȷ���ǣ�������
| A�� | �����κ�ˮ�ķ�Ӧ�����кͷ�Ӧ�����ռ���Ը����������� | |
| B�� | ����������������θ����ࣻ�ɵ�ؿ��Խ�����ת��Ϊ��ѧ�� | |
| C�� | ά���ز����ṩ�������Զ������ô�������������������Ԫ�أ�Ӧ�������� | |
| D�� | ʹ������Դ���ܼ��������������ʳ�ؽ����Σ����������õ����ţ�̽ⶾ |
14������ʵ�������ȷ���ǣ�������
| A�� | 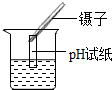 ����ҺpH | B�� | 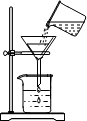 ���� | C�� | 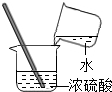 ϡ��Ũ���� | D�� | 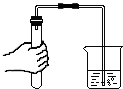 ��������� ��������� |