��Ŀ����
��A��B��C��D��E��F���ֻ�������Ƿֱ������⡢����������Ԫ���е����ֻ�������ɣ�����E������������Σ�F�Ǻ������Σ�F�����ᱵ��Һ��ϡ����Ļ����Һ��������ɫ�������������ֻ�����������ת���ϵ��
��A+B��E+H2O��D+C��F��A+C��E+H2O��B+D��F+H2O
��1���ɴ��ƶϳ������ֻ�����ķ���ʽ����ѧʽ���� A��_________��B��_________��C��_________��D��_________��E��_________��F��_________��
��2��д������������ˮ���ɵĻ�ѧ����ʽ���١�_________�� �ۡ�_________�� �ܡ�_________������
��A+B��E+H2O��D+C��F��A+C��E+H2O��B+D��F+H2O
��1���ɴ��ƶϳ������ֻ�����ķ���ʽ����ѧʽ���� A��_________��B��_________��C��_________��D��_________��E��_________��F��_________��
��2��д������������ˮ���ɵĻ�ѧ����ʽ���١�_________�� �ۡ�_________�� �ܡ�_________������
��1��H2S��NaOH��Na2O��H2SO4��Na2S��Na2SO4��
��2��H2S+2NaOH�TNa2S+2H2O��H2S+Na2O�TNa2S+H2O��H2SO4+2NaOH�TNa2SO4+2H2O��
��2��H2S+2NaOH�TNa2S+2H2O��H2S+Na2O�TNa2S+H2O��H2SO4+2NaOH�TNa2SO4+2H2O��

��ϰ��ϵ�д�
 ȫ�ܲ����ĩС״Ԫϵ�д�
ȫ�ܲ����ĩС״Ԫϵ�д�
�����Ŀ

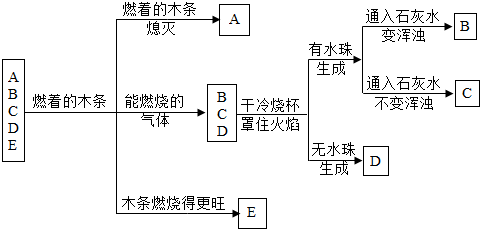

 ��A��B��C��D��E���ֳ������ʣ���������ͼ��ʾ�Ĺ�ϵ����֪��C��һ�ֽ���������D����ʹ����ʯ��ˮ����ǣ�E�����ж������������ڵ�Ѫ�쵰��ϣ��ƶ�B���ʵĻ�ѧʽ��
��A��B��C��D��E���ֳ������ʣ���������ͼ��ʾ�Ĺ�ϵ����֪��C��һ�ֽ���������D����ʹ����ʯ��ˮ����ǣ�E�����ж������������ڵ�Ѫ�쵰��ϣ��ƶ�B���ʵĻ�ѧʽ��