��Ŀ����
8��ʵ��С���о����ᡢ���������������ʵĻ�ѧ���ʣ�������ͼ��ʾ8��ʵ�飮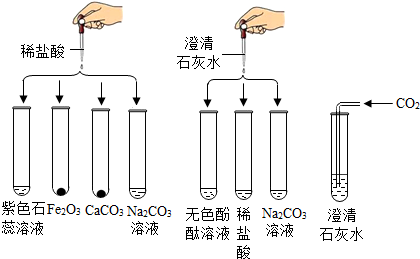
��֪��Na2CO3+CaCl2�TCaCO3��+2NaCl
��1��ʵ���ij�Թ���Ϊ��ɫ��Һ�����Թ��з�����Ӧ�Ļ�ѧ����ʽΪFe2O3+6HCl�T2FeCl3+3H2O��
��2��ʵ���ij�Թ���Ϊ��ɫ��Һ�������м������������ᣬ��Һ��Ϊ��ɫ���ɴ��ƶϣ����Թ������ʢ�е���������ɫ��̪��Һ��
��3��ʵ���ij�Թ���ֻ�õ���ɫ��Һ�������м���������Na2CO3��Һ�������������ɴ��ƶϣ����Թ������������Ӧ�Ļ�ѧ����ʽΪNa2CO3+2HCl�T2NaCl+H2O+CO2����ԭ��ɫ��Һ�е�������NaCl��д��һ�ֿ��ܼ��ɣ���
���� �������е���ͼ�������Լ����ʷ�Ӧ��ʵ��������з�����ɣ�
��� �⣺��1��ʵ���ij�Թ���Ϊ��ɫ��Һ��˵������������ϡ���ᷴӦ�������Ȼ�����ˮ������Թ��з�����Ӧ�Ļ�ѧ����ʽΪFe2O3+6HCl�T2FeCl3+3H2O�����Fe2O3+6HCl�T2FeCl3+3H2O��
��2��ʵ���ij�Թ���Ϊ��ɫ��Һ�������м���������ij��Һ���Ϊ��ɫ��˵��ԭ�Թ���ʢ�е��Ƿ�̪��Һ����������������ᣬ������ᣬ��ɫ��̪��Һ��
��3��ʵ���ij�Թ���ֻ�õ���ɫ��Һ�������м���������Na2CO3��Һ������������˵���Թ��ڵ�������̼���Ʋ���Ӧ���ɴ��ƶϣ����Թ�����̼���ƺ����ᷴӦ�������Ȼ��ơ�ˮ�Ͷ�����̼�������������Ӧ�Ļ�ѧ����ʽΪNa2CO3+2HCl�T2NaCl+H2O+CO2����ԭ��ɫ��Һ�е�������NaCl�����Ȼ��ƺ�̼���ƵĻ������Na2CO3+2HCl�T2NaCl+H2O+CO2����NaCl��
���� ���ճ�������ͼ�������Լ����ʼ䷴Ӧ��ʵ����������ȷ�����Ĺؼ���

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
18������ʵ����С�������ʵ��С���ͬѧΪ̽������ȼ�յ���������ƣ���֪�����Ż��Ϊ40�棬�����ж����а����ܹ�ȼ�յ��ǣ�������
| A�� | 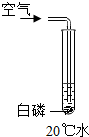 | B�� | 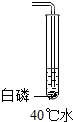 | C�� | 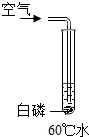 | D�� | 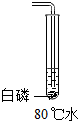 |
3���Ա��ǻ�ѧ�о�����Ҫ��������ͼ��ʾͼʾʵ���У�û�����ֶԱȷ������ǣ�������
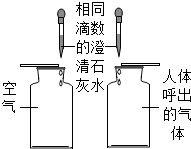 | 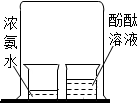 | 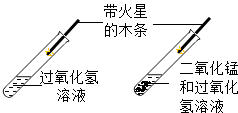 | 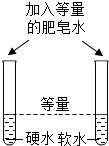 |
| A���Ƚ϶�����̼�ĺ��� | B��̽�����ӵ��˶� | C���о��������̵Ĵ����� | D������Ӳˮ����ˮ |
| A�� | A | B�� | B | C�� | C | D�� | D |
13��������Һ�У���ʹ��̪���ɫ���ǣ�������
| A�� | �ռ���Һ | B�� | ϡ���� | C�� | ϡ���� | D�� | �Ȼ�����Һ |
18����˵��ֽ��ȼ���ǻ�ѧ�仯���ǣ�������
| A�� | ���� | B�� | ���� | C�� | ���ɶ�����̼ | D�� | ֽ�ŵ������С |
 ��
��