��Ŀ����
ij�о�С����������ᾧ����Ʒ�ֽ���ﲢ�ⶨ�������������������ʲ����뷴Ӧ�������ᾧ�壨 H2C2O4•2H2O�����������ʼ�����
�۵� | �е� | ���ȶ��� | �����ʯ��ˮ��Ӧ |
101��C��102��C | 150��C��160��C���� | 100.1��Cʧȥ�ᾧˮ��175��C�ֽ��CO2��CO��H2O | ��Ca��OH��2��Ӧ������ɫ������CaC2O4�� |
��1�����ȷֽ���ᾧ�������˵�װ����____________����ͼ1��ĸ��ţ���
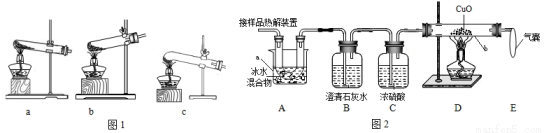
��2��ͼ2����֤�ȷֽ�����к�CO��CO2��װ��
������a��b�����Ʒֱ���____________��____________��
��֤������CO2��������________________________��֤������CO������____________��D�з�Ӧ�Ļ�ѧ����ʽ��_____________��
��װ��A��������____________________�����ҵ�������________________________��
��3��Ϊ�ⶨ��Ʒ�в��ᾧ�����������������������·�����
�ٳ�һ������Ʒ����ͼװ�ý���ʵ�飬���װ��D��Ӧǰ���������ɴ˼������ʵ������ʵ��ֵƫ�ͣ��ų������Ͳ��������أ���ԭ������У�COδ��ȫ��Ӧ��___________________________��
�ڳ�ȡ8.75g���ᾧ����Ʒ����50.00g��Һ��ȡ10.00g��Һ��������ϡ���ᣬȻ��μ�25.00g3.16%KMnO4��Һ��ǡ�÷�Ӧ��ȫ��
����֪��2KMnO4+5H2C2O4+3H2SO4=K2SO4+2MnSO4+10CO2��+8H2O����KMnO4��Һ��____________ɫ��25.00g3.16%KMnO4��Һ��KMnO4������____________g����Ʒ�в��ᾧ�����������Ϊ____________��
��Mr��H2C2O4��=90��Mr��H2C2O4•2H2O��=126��Mr��KMnO4��=158����
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�

 2X + 2Na2O + 2Na2SO4 + O2����������X�Ļ�ѧʽΪ
2X + 2Na2O + 2Na2SO4 + O2����������X�Ļ�ѧʽΪ