��Ŀ����
�������ķ�չ�벻����Դ��������ᣬ�������õ���Դ��Ҫ���Ի�ʯȼ�ϣ���1�����������������ɻ����ִ���ȼ����Ҫ�Ǵ�ʯ���и��ݲ�ͬ�е㷶Χ������������͡�����ú�͡����͵ȣ�ʯ�ͷ���Ĺ����� �仯����ʯȼ������Ҫ����̼����Ԫ�أ���ȫȼ�յIJ����� ��
��2�����������䡱�еġ�����ָ ����Щ�ط��ڹ������ͳ���������ø�������������ȼ�ϵĸĸ��������Ҫ�ɷ�ȼ�յĻ�ѧ����ʽ ��
��3��Ϊ��ʡʯ����Դ���ҹ���ʼ�ƹ㡰�����Ҵ����͡������������м������Ҵ���д���Ҵ���ȫȼ�յĻ�ѧ����ʽ ��
���𰸡���������1��ʯ���д������͡�����ú�͡����͵����ʣ�����ͨ������ķ�������Щ���ʷ��룮��ʯȼ���е�̼����Ԫ�أ���ȫȼ��ʱ���ɶ�����̼��ˮ��
��2����Ȼ������Ҫ�ɷ��Ǽ��飬ȼ�տ����ɶ�����̼��ˮ��
��3�������Ҵ����п�ȼ�ԣ���������Ӧ�����ɶ�����̼��ˮ���н��
����⣺��1��ʯ�͵ķ������û�����������ɣ����������仯����ʯȼ���е�̼Ԫ�غ���Ԫ����ȼ��ʱ�����ɶ�����̼��ˮ��
��2�������������䡱�еġ�����ָ������Ȼ������Ȼ������Ҫ�ɷ��Ǽ��飬ȼ�յĻ�ѧ��Ӧʽ�ǣ�CH4+2O2 CO2+2H20��
CO2+2H20��
��3���Ҵ��������ڵ�ȼ�����������ɶ�����̼��ˮ�����Ƿ�Ӧ�Ļ�ѧ����ʽΪ��C2H5OH+3O2 2CO2+3H2O��
2CO2+3H2O��
�ʴ�Ϊ����1��������������̼��ˮ��
��2����Ȼ����CH4����CH4+2O2 CO2+2H20��
CO2+2H20��
��3��C2H5OH+3O2 2CO2+3H2O��
2CO2+3H2O��
�����������Ƕ���Դ�������Ŀ��飬��Ҫ�Ƕ�������Դ��֪ʶ�����˷���������д��ѧ����ʽʱ������ȷ����Ӧԭ����Ȼ��������ԭ���ҳ���Ӧ�������ͷ�Ӧ���������ݷ���ʽ����д������д����ʽ��
��2����Ȼ������Ҫ�ɷ��Ǽ��飬ȼ�տ����ɶ�����̼��ˮ��
��3�������Ҵ����п�ȼ�ԣ���������Ӧ�����ɶ�����̼��ˮ���н��
����⣺��1��ʯ�͵ķ������û�����������ɣ����������仯����ʯȼ���е�̼Ԫ�غ���Ԫ����ȼ��ʱ�����ɶ�����̼��ˮ��
��2�������������䡱�еġ�����ָ������Ȼ������Ȼ������Ҫ�ɷ��Ǽ��飬ȼ�յĻ�ѧ��Ӧʽ�ǣ�CH4+2O2
 CO2+2H20��
CO2+2H20����3���Ҵ��������ڵ�ȼ�����������ɶ�����̼��ˮ�����Ƿ�Ӧ�Ļ�ѧ����ʽΪ��C2H5OH+3O2
 2CO2+3H2O��
2CO2+3H2O���ʴ�Ϊ����1��������������̼��ˮ��
��2����Ȼ����CH4����CH4+2O2
 CO2+2H20��
CO2+2H20����3��C2H5OH+3O2
 2CO2+3H2O��
2CO2+3H2O�������������Ƕ���Դ�������Ŀ��飬��Ҫ�Ƕ�������Դ��֪ʶ�����˷���������д��ѧ����ʽʱ������ȷ����Ӧԭ����Ȼ��������ԭ���ҳ���Ӧ�������ͷ�Ӧ���������ݷ���ʽ����д������д����ʽ��

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
�������ķ�չ�벻����Դ��������ᣬ�������õ���Դ��Ҫ���Ի�ʯȼ�ϣ�
��1�����������������ɻ����ִ���ȼ����Ҫ�Ǵ�ʯ���и��ݲ�ͬ�е㷶Χ������������͡�����ú�͡����͵ȣ�ʯ�ͷ���Ĺ����� �仯����ʯȼ������Ҫ����̼����Ԫ�أ���ȫȼ�յIJ����� ������������Ԫ�أ�ȼ��ʱ��ת��Ϊ ��
��2���������䣬���ı����о��������ȼ�Ͻṹ���±��Ǽ�������ȼ�ϵļ۸����ֵ��
�ۺϿ��ǣ��㽫ѡ������ȼ�� ������ ��
��3����ʯȼ�ϲ��������ʵ�������Դ�����DZ���Ļ�����Դ������ϩ��һ����õ����ϣ�������ʯ���ѽ�������ϩ��CH2=CH2��Ϊԭ�ϣ���������ϩ���Ӵ�˫����-CH2-CH2-������һ�𣬾ۺϳɴ���Ӷ��γɵģ���ͼ�Ǿ���ϩ�����γɹ��̵�ʾ��ͼ����ͨ���е���С�˴���һ����ϩ���ӣ�����ϩ���еIJ��ֽṹ�ɱ�ʾΪ���� ����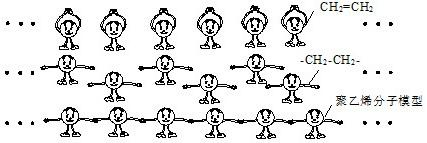
��4����������������Ч������֮���ߵ�����Դ����21�����������ٵ���Ҫ���⣮
��2006��1������ʡ�ձ������ƹ�ʹ���Ҵ����ͣ��Ҵ�������ũ������Ƶã�ũ�����ڴ���������ת��Ϊ�����ǣ�C6H12O6����Ȼ���������ھƻ�ø��������ת��Ϊ�Ҵ��Ͷ�����̼��д��������ת��Ϊ�Ҵ��Ļ�ѧ����ʽ ��
��������һ��δ���������Դ����ͨ������Ƶõ������л���CO2��O2��ˮ�����ȣ��������������ͨ��Ϊ60%��90%����Ϊ�˵õ��������������������������ͨ��NaOH��Һ�����ȵ�Cu˿����װ�ã��ó��ӷ����Ƿ���ȷ�� �������� ��
��1�����������������ɻ����ִ���ȼ����Ҫ�Ǵ�ʯ���и��ݲ�ͬ�е㷶Χ������������͡�����ú�͡����͵ȣ�ʯ�ͷ���Ĺ�����
��2���������䣬���ı����о��������ȼ�Ͻṹ���±��Ǽ�������ȼ�ϵļ۸����ֵ��
| ȼ���� | ú�� | Һ��ʯ���� | ��Ȼ�� |
| �۸� | 1.4Ԫ/ǧ�� | 6.0Ԫ/ǧ�� | 1.8Ԫ/ǧ�� |
| ��ֵ | 1.56��104kJ/kg | 4.5��104kJ/kg | 3.9��104kJ/kg |
��3����ʯȼ�ϲ��������ʵ�������Դ�����DZ���Ļ�����Դ������ϩ��һ����õ����ϣ�������ʯ���ѽ�������ϩ��CH2=CH2��Ϊԭ�ϣ���������ϩ���Ӵ�˫����-CH2-CH2-������һ�𣬾ۺϳɴ���Ӷ��γɵģ���ͼ�Ǿ���ϩ�����γɹ��̵�ʾ��ͼ����ͨ���е���С�˴���һ����ϩ���ӣ�����ϩ���еIJ��ֽṹ�ɱ�ʾΪ����

��4����������������Ч������֮���ߵ�����Դ����21�����������ٵ���Ҫ���⣮
��2006��1������ʡ�ձ������ƹ�ʹ���Ҵ����ͣ��Ҵ�������ũ������Ƶã�ũ�����ڴ���������ת��Ϊ�����ǣ�C6H12O6����Ȼ���������ھƻ�ø��������ת��Ϊ�Ҵ��Ͷ�����̼��д��������ת��Ϊ�Ҵ��Ļ�ѧ����ʽ
��������һ��δ���������Դ����ͨ������Ƶõ������л���CO2��O2��ˮ�����ȣ��������������ͨ��Ϊ60%��90%����Ϊ�˵õ��������������������������ͨ��NaOH��Һ�����ȵ�Cu˿����װ�ã��ó��ӷ����Ƿ���ȷ��

