��Ŀ����
��������ԭ����ͭ�ķ����ⶨij����ͭ��Ʒ�Ĵ��ȣ����ʲ���������Ӧ����ͬѧ�����������ʵ��װ�ã�ȷ��ȡ15�˸�����Ʒ����ʵ�飮

��1����ʵ���У���װ�õ������� ��
��2��ȷ��ȡ��ȫ��Ӧǰ��װ�ñ��Ͷ������������
С������װ�ñ����ٵ�����������Ԫ���������������ͭ������Ϊ �ˣ�����Ϊ ��С�������װ�ö��������仯���м��㣬���С���������ͭ��Ʒ�Ĵ��ȣ�д��������̣���

��1����ʵ���У���װ�õ�������
��2��ȷ��ȡ��ȫ��Ӧǰ��װ�ñ��Ͷ������������
| װ�ñ� | װ�ö� | |
| ��Ӧǰ | 48.8�� | 161.2�� |
| ��Ӧ�� | 46.5�� | 163.9�� |
���㣺ʵ��̽�����ʵ���ɳɷ��Լ�����,����ľ��������ӣ�,����ĸ����ˮ��,���ݻ�ѧ��Ӧ����ʽ�ļ���,�����Ļ�ѧ������ȼ��ʵ��
ר�⣺��ѧ̽��
��������1��������ˮ����ͭ����ˮ���������Կ��ǣ�
��2���������������������ͭ����Ԫ�ص�������������Ԫ�ص�������������ͭ���������ټ��㴿�ȣ�����Ũ���������ˮ�ԣ�����Ũ�����������������������ɵ�ˮ���������ٸ���ˮ���������������ͭ���������ټ��㴿�ȣ�
��2���������������������ͭ����Ԫ�ص�������������Ԫ�ص�������������ͭ���������ټ��㴿�ȣ�����Ũ���������ˮ�ԣ�����Ũ�����������������������ɵ�ˮ���������ٸ���ˮ���������������ͭ���������ټ��㴿�ȣ�
����⣺��1������ͨ��Ũ������Ϊ�˳�ȥ�����е�ˮ�֣���ˮ����ͭ����ˮ�ɰ�ɫ����������ͨ����ˮ����ͭ���Ǽ���Ũ�����Ƿ��������ˮ�ֳ�����
��2���з���ʽH2+CuO
Cu+H2O����֪�����ٵ�����������Ԫ������������ͭ����Ԫ�ص�����Ϊ��48.8g-46.5g=2.3g����������ͭ������Ϊ
=11.5g������ͭ����������Ϊ
��100%=76.7%��
����Ũ�����������������������ɵ�ˮ����������������ˮ������Ϊ��163.9g-161.2g=2.7g����Ҫ����2.7gˮ��Ҫ�μӷ�Ӧ������ͭ������Ϊx��
H2+CuO
Cu+H2O
80 18
x 2.7g
���ݣ�
=
���x=12g����������ͭ������������
��100%=80%
������ͭ��Ʒ�Ĵ���Ϊ80%��
�ʴ�Ϊ����1������������ˮ���Ƿ�ȫ�������գ���2��11.5��76.7��80%��
��2���з���ʽH2+CuO
| ||
| 2.3g | ||
|
| 11.5g |
| 15g |
����Ũ�����������������������ɵ�ˮ����������������ˮ������Ϊ��163.9g-161.2g=2.7g����Ҫ����2.7gˮ��Ҫ�μӷ�Ӧ������ͭ������Ϊx��
H2+CuO
| ||
80 18
x 2.7g
���ݣ�
| 80 |
| x |
| 18 |
| 2.7g |
| 12g |
| 15g |
������ͭ��Ʒ�Ĵ���Ϊ80%��
�ʴ�Ϊ����1������������ˮ���Ƿ�ȫ�������գ���2��11.5��76.7��80%��
�������������׳����ĵط��Ƕ�����������ͬ��ԭ�������Ҫ����Ũ�������ˮ�ԣ��������������ɵ�ˮ���������е�ˮҲ�����ˣ�

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
 ��ͼ�Ǽס����������ʣ������ᾧˮ�����ܽ�����ߣ�����˵������ȷ���ǣ�������
��ͼ�Ǽס����������ʣ������ᾧˮ�����ܽ�����ߣ�����˵������ȷ���ǣ�������| A�������к���������ʱ�������ý��½ᾧ�ķ����ᴿ�� |
| B��t2��ʱ�ı�����Һ������t1���Ϊ��������Һ |
| C��t1��ʱ��50g�ı�����Һ����15g�� |
| D��t2��ʱ���������ļס�����Һ����������������� |

 ����
���� �еĻ�ѧʽ��
�еĻ�ѧʽ��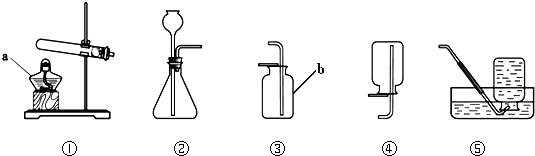

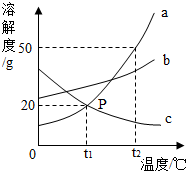 ��ͼ��a��b��c�������ʵ��ܽ�����ߣ�a��c���ܽ�������ཻ��P�㣮��ͼ�ش�
��ͼ��a��b��c�������ʵ��ܽ�����ߣ�a��c���ܽ�������ཻ��P�㣮��ͼ�ش�