��Ŀ����
13����1����ͼ1��ʾ��ij��ˮ������Ȼ��Ϊȼ��
��д����Ȼ����ȫȼ�յĻ�ѧ����ʽCH4+2O2$\frac{\underline{\;��ȼ\;}}{\;}$CO2+H2O
�ڵ����������ױ����ֶ�ס��ȼ�ս������ж�����M����M�Ļ�ѧʽCO
��2��1Kg��ͬȼ��ȼ�ղ�����CO2��SO2���������ʾ
�����н�����ȼ������Ȼ��ʹ��ú ��ȼ��ʱ�����ײ������ꣻ
�������γɹ�����ijһ���ķ�Ӧ����ʾ��ͼ��ͼ2��ʾ
| ȼ�� | ȼ�ղ�������/g | |
| CO2 | SO2 | |
| ���� | 2900 | 5.0 |
| ��Ȼ�� | 2500 | 0.1 |
| ú | 2500 | 11.0 |
���� �������е����ʵ����ʽ��з�����𣬼���ȼ�����ɵ��Ƕ�����̼��ˮ�����ݺ�̼���ʲ���ȫȼ�ջ�����һ����̼��𣻸��ݱ����ƶ�����ȼ�Ϻ�����������ȼ�ϣ�������ģ��ʾ��ͼд���йط�Ӧ�Ļ�ѧ����ʽ�����ж����ʵ������ȣ�
��� �⣺��1���ټ���ȼ�����ɶ�����̼��ˮ����Ӧ�ķ���ʽ�ǣ�CH4+2O2$\frac{\underline{\;��ȼ\;}}{\;}$CO2+2H2O��
�ں�̼���ʲ���ȫȼ�ջ�����һ����̼��һ����̼���ж������壻
��2���ٸ��ݱ�����Կ�����������ȼ������Ȼ����úȼ���ͷŵĶ���������࣬�����γ����ꣻ
�ھ�ͼ���Կ������÷�Ӧ�Ƕ���������������´����������·�Ӧ��������������Ӧ�ķ���ʽ�ǣ�2SO2+O2$\frac{\underline{\;\;\;����\;\;\;}}{����}$2SO3��
�ʴ�Ϊ����1����CH4+2O2$\frac{\underline{\;��ȼ\;}}{\;}$CO2+H2O�� ��CO����2������Ȼ���� ú����2SO2+O2$\frac{\underline{\;\;\;����\;\;\;}}{����}$2SO3��
���� ���⿼����ǻ�ѧ����Դ��֪ʶ����ɴ��⣬�����������е�֪ʶ�������ṩ����Ϣ���У�

��ϰ��ϵ�д�
 ���Ͱ�ͨ��ĩ���ϵ�д�
���Ͱ�ͨ��ĩ���ϵ�д�
�����Ŀ
3��ij�Ͻ�12g��������ϡ�����ַ�Ӧ���������0.4g�������úϽ��е�Ԫ�ؿ����ǣ�������
| A�� | Mg��Al | B�� | Zn��Fe | C�� | Zn��Cu | D�� | Cu��Ag |
1�������йؽ�����˵����ȷ���ǣ�������
| A�� | �����£����������Ժ�������Ӧ | B�� | �������ܺ��ᷴӦ�������� | ||
| C�� | ������Ǵ��� | D�� | ��ͭ��Ӳ�ȱȴ�ͭ�� |
8�����и������ʣ�ǰ�����ڻ����������ڻ�������ǣ�������
| A�� | ��ˮ����� Һ�� | B�� | ���� �� | ||
| C�� | ����ˮ Һ�� | D�� | ���� �� |
18���������������������أ�������Ʒ���õIJ��ϲ����ڽ������ϵ��ǣ�������
| A�� | �ѺϽ������ | B�� | ����̤���ࡱ | C�� | ��ʯ | D�� | �ƽ���Ʒ |
5����ͼ��ʾʵ�����������ȷ���ǣ�������
| A�� |  Ϩ��ƾ��� | B�� | 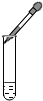 �μ�Һ�� | C�� | 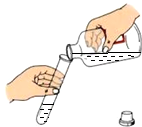 �㵹Һ�� | D�� | 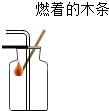 �������� �������� |
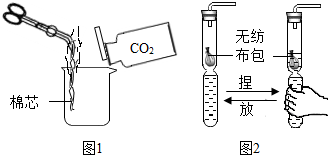 ��Ƕ���ʶ���ʣ��ܰ������Ǹ�ȫ���˽��������磮�������Ͷ�����̼Ϊ�����ش��������⣺
��Ƕ���ʶ���ʣ��ܰ������Ǹ�ȫ���˽��������磮�������Ͷ�����̼Ϊ�����ش��������⣺