��Ŀ����
Ӳˮ������ǵ���������������ܶ�Σ��������Ӳˮϴ������˷ѷ�����Ҳϴ��������չ�¯ʱ��Ӳˮ��ʹ��¯�ڽ�ˮ���������˷�ȼ�ϣ�����ʱ��������ը���˳�������Ӳˮ���ܻ�ý�ʯ��
a) �����ܽ���ˮ��Ӳ�ȷ����� ������ţ���
�ٹ��� �ڼӻ���̿ ����� �ܼ�������
b) ��ѧУ��չ�о���ѧϰʱ��ijͬѧ���������ϡ�����ȥˮ������Ҫ�ɷ���Mg(OH)2��CaCO3������д����صĻ�ѧ��Ӧ����ʽ��
___________________________________��___________________________________��
�� Mg(OH)2+ 2HCl = MgCl2 + H2O CaCO3 + 2HCl = CaCl2 + H2O + CO2��

������ߺ�DZˮͧ�п��ù������ƣ�Na2O2������������Ϊ��̽���䷴Ӧԭ������ȤС��ͬѧ�ڽ�ʦ��ָ���£�����������̽����
�����ʵ��� С��ͬѧ��ͨ����ͼ��ʾװ��̽�����������������̼�ķ�Ӧ������֤��Ӧ���
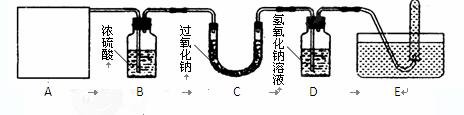
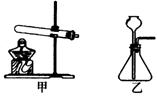
(1) A�Ƕ�����̼����ķ���װ�ã�Ӧѡ����ͼ�е�_________����ס����ҡ���װ�á�ʵ������ȡ������̼�Ļ�ѧ����ʽΪ________________________________________��
(2) ����Eװ���Թ����ռ��������壬��E���Թ������ռ������Թ��Ƴ�ˮ�棬���Թ�������ȼ�յ�ľ��������_______________��֤�����ɵ�ΪO2��
(3) B��Ũ�����������________________________________________________________��
(4) ����C�з�Ӧ��ʣ�����ijɷ֣�
| ʵ�鲽�� | ʵ������ | ʵ����� |
| ��ȡ����C�з�Ӧ��Ĺ������Թ��� �����Թ��еμ�����ϡ���� ��_____________ | a________________ b��Һ����� | ��Ӧ�����ɵĹ�����̼���� |
(5) ��д���������ƣ�Na2O2���������̼��Ӧ�ķ���ʽ��
_____________________________________________________________________��
 ��
�� ���¼�����
���¼�����  �C131��53�ŵ�Ԫ�ص�һ��ԭ�ӣ���� �C131�ĺ˵����Ϊ�� ��
�C131��53�ŵ�Ԫ�ص�һ��ԭ�ӣ���� �C131�ĺ˵����Ϊ�� �� ��Һ���������������γ���ɫ��
��Һ���������������γ���ɫ�� ����
����