��Ŀ����
2��������ѧ֪ʶ�ش��������⣺��1��Ũ������лӷ��ԣ���Ũ���������ˮ�ԣ���Ũ�����Ũ���᳨�ڷ���һ��ʱ�䣬�����������������С�������С�����䡱����
��2����һ���������������ϡ�����У��ȹ۲쵽�������ܽ⣬��Һ����ɫ��Ϊ��ɫ����һ������ɹ۲쵽�������ݵ������漰���Ļ�ѧ����ʽΪ��
��Fe2O3+6HCl=2FeCl3+3H2O��
��2HCl+Fe=FeCl2+H2����
���� ��1������Ũ������лӷ��ԣ���Ũ���������ˮ�Խ��н��
��2���������Ҫ�ɷ������������������������ᷴӦ�����Ȼ�����ˮ���Ȼ�����ˮ��Һ�ʻ�ɫ�������ȥ���������ᷴӦ�����Ȼ��������������ݴ˽��
��� �⣺��1��Ũ������лӷ��ԣ��ӷ����Ȼ������壬��Ũ���������ˮ�ԣ�����Ũ�����Ũ���᳨�ڷ���һ��ʱ�䣬�����������������С��
��2���������Ҫ�ɷ������������������������ᷴӦ�����Ȼ�����ˮ���Ȼ�����ˮ��Һ�ʻ�ɫ�������ȥ���������ᷴӦ�����Ȼ��������������漰���Ļ�ѧ����ʽΪ��Fe2O3+6HCl�T2FeCl3+3H2O��Fe+2HCl�TFeCl2+H2����
�ʴ�Ϊ��
��1���ӷ��ԣ� ��ˮ�ԣ� ��С��
��2���������ܽ⣬��Һ����ɫ��Ϊ��ɫ
��Fe2O3+6HCl=2FeCl3+3H2O
��2HCl+Fe=FeCl2+H2��
���� ���⿼���˳������ʵ����ʣ���ɴ��⣬�����������е�֪ʶ���У�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
12�����ʱ���ķ���ȡ�������ʣ�����ҩƷ����Ҫ�ܷⱣ����ǣ�������
| A�� | ����ʯ��ˮ | B�� | Ũ���� | C�� | �Ȼ��� | D�� | Ũ���� |
7��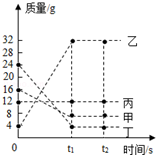 ���ܱ������м����������ʣ���һ�������·�����ѧ��Ӧ����÷�Ӧǰ��t1��t2ʱ������������ͼ��ʾ������˵���в���ȷ���ǣ�������
���ܱ������м����������ʣ���һ�������·�����ѧ��Ӧ����÷�Ӧǰ��t1��t2ʱ������������ͼ��ʾ������˵���в���ȷ���ǣ�������
 ���ܱ������м����������ʣ���һ�������·�����ѧ��Ӧ����÷�Ӧǰ��t1��t2ʱ������������ͼ��ʾ������˵���в���ȷ���ǣ�������
���ܱ������м����������ʣ���һ�������·�����ѧ��Ӧ����÷�Ӧǰ��t1��t2ʱ������������ͼ��ʾ������˵���в���ȷ���ǣ�������| A�� | �÷�ӦΪ���Ϸ�Ӧ | |
| B�� | ������Ϊ�÷�Ӧ�Ĵ��� | |
| C�� | �÷�Ӧ�У��μӷ�Ӧ�ļס���������֮��Ϊ2��5 | |
| D�� | �÷�Ӧ�У����ɵ�������Ϊ32g |
11�������±������ش����⣮
��1��20��KNO3���ܽ����31.6g��
��2��60��ʱ��200gKNO3��Һ�к���100g��������Һ������20�棬������KNO368.4g��
��3��NaCl��Һ�к�������KNO3���ᴿNaCl�����õķ����������ᾧ��������½ᾧ���������ᾧ������
| �¶�/�� | 0 | 20 | 40 | 60 | 80 | 100 | |
| �ܽ��/g | KNO3 | 13.3 | 31.6 | 63.9 | 110 | 169 | 246 |
| NaCl | 25.7 | 36.0 | 36.6 | 37.3 | 38.4 | 39.8 | |
��2��60��ʱ��200gKNO3��Һ�к���100g��������Һ������20�棬������KNO368.4g��
��3��NaCl��Һ�к�������KNO3���ᴿNaCl�����õķ����������ᾧ��������½ᾧ���������ᾧ������
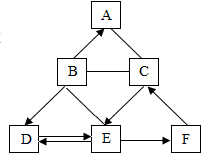 ͼ��A��FΪ���л�ѧ�������������ʣ�����A��B��D����ͬ�������ӣ�C��D��E��F����ͬһ��Ԫ�أ�C��E��F�Dz�ͬ�������ʣ�A��C֮��ķ�Ӧ�����ڼ����̬���ʣ�B��E֮��ķ�Ӧ��ʵ������ȡ������̼�ķ�Ӧԭ������ͼ�С�-����ʾ�������������ʼ��ܷ�����ѧ��Ӧ����������ʾ���ʼ����ת����ϵ�����ַ�Ӧ������P��Ӧ��������ȥ�����ش��������⣺
ͼ��A��FΪ���л�ѧ�������������ʣ�����A��B��D����ͬ�������ӣ�C��D��E��F����ͬһ��Ԫ�أ�C��E��F�Dz�ͬ�������ʣ�A��C֮��ķ�Ӧ�����ڼ����̬���ʣ�B��E֮��ķ�Ӧ��ʵ������ȡ������̼�ķ�Ӧԭ������ͼ�С�-����ʾ�������������ʼ��ܷ�����ѧ��Ӧ����������ʾ���ʼ����ת����ϵ�����ַ�Ӧ������P��Ӧ��������ȥ�����ش��������⣺ 1946���һ̨�������������������60�����ʱ�䣬��չ�dz�Ѹ�ͣ����Ѿ��߽��칫�ң��߽���ͥ����ͼ��ij�칫���õļ�����ʹ�ӡ������ش��й����⣺
1946���һ̨�������������������60�����ʱ�䣬��չ�dz�Ѹ�ͣ����Ѿ��߽��칫�ң��߽���ͥ����ͼ��ij�칫���õļ�����ʹ�ӡ������ش��й����⣺