��Ŀ����
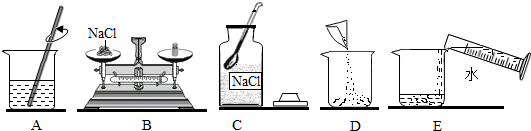
9����1����֪��ˮ���ܶ�Ϊ1.0g/mL��ʵ��������50g������������Ϊ5%���Ȼ�����Һ����Ҫ��ȡ�Ȼ���2.5g����Ҫ��ȡˮ47.5mL����2�����ƵIJ���ʾ��ͼ���£���ʵ�����ȷ����˳��ΪCBDEA������ĸ��ţ���
��3������⣬��������Һ��������������ƫС����ԭ������Ǣ٢ڢۣ�����ţ���
���Ȼ��ƹ��岻������������������ �ڳ���ʱ�����������������ͬ��ֽƬ
����ȡˮʱ�����Ӷ������������� ��װƿʱ����������Һ������
���� ��1��������������=��Һ���������ʵ������������ٸ����ܼ�����=��Һ����-�������������������ˮ��������
��2���������е���Һ���Ƶ�֪ʶ���з�����𣬸�����Һ���ƵIJ�����
��3��������������ƫС���������ʼ�С�����ܼ������Ե�ʣ��ݴ˽��
��� �⣺
��1��������������=��Һ���������ʵ�������������������5%����ˮ50g����Ҫ��ȡNaCl����Ϊ50g��5%=2.5g��
�ܼ�����=��Һ����-����������������ˮ������=50g-2.5g=47.5g����ˮ�����Ϊ��$\frac{47.5g}{1g/mL}$=47.5mL��
��2��������Һʱ�����ȴ��Լ�ƿ��ȡʳ�ι��壬Ȼ�������ƽ�ϳ������ٷŽ��ձ��У�Ȼ����ȡ�õ�ˮ�����ձ��У�����ò����������ܽ⣬���CBDEA��
��3�����Ȼ��ƹ��岻��������ʳ�ε�����ƫС����������������С��
�ڳ���ʱ�����������������ͬ��ֽƬ����ҩƷ��ʳ�ε���������ֽƬ��������������˵�������ʳ�ε�������С����������������С��
����ȡˮʱ�����Ӷ���������ˮ�࣬��������������С��
��װƿʱ����������Һ��������Ӱ����������������
����٢ڢۣ�
�𰸣�
��1��47.5mL��
��2��CBDEA��
��3���٢ڢۣ�
���� ���⿼�������Һ�����ƣ���ɴ��⣬�����������е���Һ���ƵIJ����Լ������������������ı�����ؽ��У�

| A�� | ij�������ʼ���ϡ�����ܲ������壬��ù�������ǻ��ý��� | |
| B�� | ��ˮֱͨ������Բ�����������������ˮ����������������ɵ� | |
| C�� | ͬ��Ԫ�ص�ԭ����������ͬ������������ͬ��������ͬ��Ԫ�� | |
| D�� | �кͷ�Ӧ�IJ������κ�ˮ���������κ�ˮ�ķ�Ӧһ�����кͷ�Ӧ |
| A�� | ������ | B�� | ������ | C�� | ������ | D�� | ͭ���� |
| A�� | ���ʯ��Ӳ����ʯīҲ��Ӳ | |
| B�� | �����ж��������������꣬���Կ����ж�����̼һ����������� | |
| C�� | �����Ǵ���ɵ��������Դ���ɵ���һ�������� | |
| D�� | ͬ���·ֽ�����أ��Ӵ����ķ�Ӧ���ʿ죬�ʴ������Ըı䷴Ӧ���� |
��1��������Ϊ�����������ṩ�������������������һ�֣����������ڷ����ķ�Ӧ�ǣ�C6H12O6+6O2�T6CO2+6X����X�Ļ�ѧʽΪH2O��
��2���п��ٽ��������С���ƶ�������ʳ�ף����л�ȱ��һ����������Ӫ���أ�
| ��ʳ | ������ͷ |
| ��ʳ | ����顢��Ѽ���������⡢������ |
| ���� | ţ�� |
��3��ʵ������װ������������Һ���Լ�ƿ�����ò�������ԭ�����ڳ����£�NaOH�벣�����е�SiO2�����ط�����Ӧ����Na2SiO3��H2O��Na2SiO3ʹƿ����ƿ��ճ����һ�����ʹ�ã��÷�Ӧ�Ļ�ѧ����ʽΪ2NaOH+SiO2=Na2SiO3+H2O��
| A�� | ����Һ��ʹ��ɫʯ���죬����ʹ��ɫʯ����Һ����һ��������Һ | |
| B�� | �������Ԫ�غ���Ԫ�أ����Ժ�����Ԫ�غ���Ԫ�ص�����һ���Ǽ� | |
| C�� | �����pHС��7������pHС��7����ˮ������ | |
| D�� | ������̼�����������Ʒ�Ӧ�����κ�ˮ���������̼Ҳ�����������Ʒ�Ӧ�����κ�ˮ |
 ͼ1��Ԫ�����ڱ�����Ԫ�صIJ�����Ϣ��ͼ2����ԭ�ӵĽṹʾ��ͼ������˵���У�����ȷ����B
ͼ1��Ԫ�����ڱ�����Ԫ�صIJ�����Ϣ��ͼ2����ԭ�ӵĽṹʾ��ͼ������˵���У�����ȷ����B