��Ŀ����
a����ʵ������ȼƷ��������ƿʧȥ��ǩ���Լ���ijͬѧΪ̽����ɷ֣���������ʵ�������ȡ2.16g�����ʣ�ʹ����ȫȼ�գ���ֻ֪����2.16gˮ��5.28g������̼������������գ�
��1���������к���Ԫ��______ g����̼Ԫ��______g��
��2����������______ �������������������Ԫ�أ���������Ԫ�أ�����Ԫ������Ϊ______ g����������Ԫ�أ��˿ղ����
b�����������ػ�ѧʽΪC12H10O5���ǻ�ɫ��Ȼ�ɫ�ᾧ��ĩ������������ˮ������Ϊ��������C3H8O2�����ܳ���������ٴ�����ҩΪ�����⾷ҩ�������ڼ��Ե����ס����Ե������Է���������dz����θ�ȣ��Լ��㣺
��1�����������ء�����Է�������Ϊ______��
��2�����������ء���C��H��O����Ԫ�ص�������Ϊ______��
��3��78g���������ء���ȫȼ��ʱ��������______g��
�⣺a�����ݷ�������֪��ˮ����Ԫ�غͶ�����̼�е�̼Ԫ��ȫ�������ڸ�δ֪�������У��ʿ��������Ԫ�غ�̼Ԫ�ص���������Ԫ�ص�����=2.16g��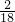 ��100%=0.24g��̼Ԫ�ص�����=5.28g��
��100%=0.24g��̼Ԫ�ص�����=5.28g��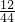 ��100%=1.44g������1.44g+0.24g=1.68g��2.16g�����������غ㶨�ɿ���֪���������к�����Ԫ�أ�������Ϊ��2.16g-1.68g=0.48g��
��100%=1.44g������1.44g+0.24g=1.68g��2.16g�����������غ㶨�ɿ���֪���������к�����Ԫ�أ�������Ϊ��2.16g-1.68g=0.48g��
b����1�����ʵ���Է����������ڸ�ԭ�ӵ����ԭ������������ԭ�Ӹ������ʡ��������ء�����Է�������Ϊ��12��12+1��10+16��5=234��
��2���������и�Ԫ�ص�������Ϊ����12��12������1��10������16��5��=72��5��40��
��3������������������Ϊx����
C12H10O5+12O2 12CO2+5H2O
12CO2+5H2O
234 384
78g x
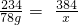
��ã�x=128g��
�ʴ�Ϊ��a��0.24g��1.44g������0.48g��
b����1��234��
��2��72��5��40��
��3��128g��
������a������Ԫ�ص�����������������������⡢��Ԫ�ص�������Ȼ�����������غ㶨�����ж��Ƿ�����Ԫ�أ��������������
b����1�����ݻ�ѧʽ�������Է���������
��2��Ԫ�ص������ȵ��������ԭ�������͵ıȣ�
��3�����ݻ�ѧ����ʽ�������������������
�������ܹ����������������غ㶨���������ʵijɷֽ����жϣ����ܹ����ݻ�ѧ����ʽ���м��㣮
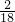 ��100%=0.24g��̼Ԫ�ص�����=5.28g��
��100%=0.24g��̼Ԫ�ص�����=5.28g��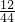 ��100%=1.44g������1.44g+0.24g=1.68g��2.16g�����������غ㶨�ɿ���֪���������к�����Ԫ�أ�������Ϊ��2.16g-1.68g=0.48g��
��100%=1.44g������1.44g+0.24g=1.68g��2.16g�����������غ㶨�ɿ���֪���������к�����Ԫ�أ�������Ϊ��2.16g-1.68g=0.48g��b����1�����ʵ���Է����������ڸ�ԭ�ӵ����ԭ������������ԭ�Ӹ������ʡ��������ء�����Է�������Ϊ��12��12+1��10+16��5=234��
��2���������и�Ԫ�ص�������Ϊ����12��12������1��10������16��5��=72��5��40��
��3������������������Ϊx����
C12H10O5+12O2
 12CO2+5H2O
12CO2+5H2O234 384
78g x
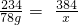
��ã�x=128g��
�ʴ�Ϊ��a��0.24g��1.44g������0.48g��
b����1��234��
��2��72��5��40��
��3��128g��
������a������Ԫ�ص�����������������������⡢��Ԫ�ص�������Ȼ�����������غ㶨�����ж��Ƿ�����Ԫ�أ��������������
b����1�����ݻ�ѧʽ�������Է���������
��2��Ԫ�ص������ȵ��������ԭ�������͵ıȣ�
��3�����ݻ�ѧ����ʽ�������������������
�������ܹ����������������غ㶨���������ʵijɷֽ����жϣ����ܹ����ݻ�ѧ����ʽ���м��㣮

��ϰ��ϵ�д�
 �̲�ȫ���ִʾ�ƪϵ�д�
�̲�ȫ���ִʾ�ƪϵ�д�
�����Ŀ
��ʵ������ȼƷ��������һƿʧȥ��ǩ���Լ���ij��ѧ��ȤС��ͬѧΪ̽������ɣ�����������ʵ�顣
��1����ͬѧȡ�����������������������г��ȼ�գ���������������ˮ�Ͷ�����̼���ɴ˿ɵó��Ľ�����
��ѡ����ţ���
| A����������ֻ����̼����Ԫ�� |
| B����������ֻ����̼����Ԫ�� |
| C����������һ������̼����Ԫ�أ����ܺ�����Ԫ�� |
| D����������̼���⡢������Ԫ����� |
��2����ͬѧȡ3.6g�������������������г��ȼ�գ����ⶨ����5.4gˮ��11g������̼��5.4gˮ�к���Ԫ�� g��11g������̼�к�̼Ԫ�� g����������� ��Ԫ�أ�ѡ����С�����������
��3����ͬѧ������ͬѧ��ʵ�����ݺͷ����������һ���������������̼ԭ�Ӻ���ԭ�ӵĸ�����Ϊ ������������ȣ���