��Ŀ����
13�������������г�����һ�ּ�ϸС��CaCO3��ĩ��ΪĦ����������ʯ��ʯΪԭ�Ͻ���������ʯ��ʯ�����ʣ����ʲ����뷴ӦҲ������ˮ������ͼ��ʵ����ģ��������Ħ������������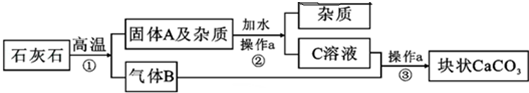
��1��ʵ�����a�������ǹ��ˣ�
��2��д��Ӧ�ٵĻ�ѧ����ʽ��CaCO3$\frac{\underline{\;����\;}}{\;}$CaO+CO2����
��3��������д�������B�����������������ЧӦ��
��4��д����Ӧ�۵Ļ�ѧ����ʽ��Ca��OH��2+CO2�TCaCO3��+H2O��
��4�����ڿ�״CaCO3�ͷ�ĩ״CaCO3�зֱ����ϡ���ᣬ�ų����������Ƿ�ĩ״̼��ƣ������Ƿ�Ӧ��ĽӴ������
���� ��1�����ݹ��˿��Խ������Һ�������з�����
��2������̼����ڸ��µ������·�Ӧ���������ƺͶ�����̼���з�����
��3�����ݶ�����̼����������ЧӦ���з�����
��4�����ݶ�����̼���������Ʒ�Ӧ����̼��Ƴ�����ˮ���з�����
��5�����ݷ�Ӧ�������뷴Ӧ��ĽӴ�������¶ȡ���Ӧ���Ũ���йؽ��з�����
��� �⣺��1����������a�õ�����ҺC�����ʣ����Բ���a�ǹ��ˣ�
��2��̼����ڸ��µ������·�Ӧ���������ƺͶ�����̼����ѧ����ʽΪ��CaCO3$\frac{\underline{\;����\;}}{\;}$CaO+CO2����
��3��������̼����������ЧӦ��
��3��������̼���������Ʒ�Ӧ����̼��Ƴ�����ˮ����ѧ����ʽΪ��Ca��OH��2+CO2�TCaCO3��+H2O��
��4���ڿ�״CaCO3�ͷ�ĩ״CaCO3�зֱ����ϡ���ᣬ�ų����������Ƿ�ĩ״̼��ƣ������ǣ���Ӧ��ĽӴ������
�ʴ�Ϊ����1�����ˣ�
��2��CaCO3$\frac{\underline{\;����\;}}{\;}$CaO+CO2����
��3�����ң�
��4��Ca��OH��2+CO2�TCaCO3��+H2O��
��5����ĩ״̼��ƣ���Ӧ��ĽӴ������
���� �ڽ������ʱ�����ȷ������п�������⣬Ȼ�������еķ�Ӧ���̺�ѧ����֪ʶ���н��

 ������ѧ���̲���ȫ���ϵ�д�
������ѧ���̲���ȫ���ϵ�д� ������ʱ����ҵ����ϵ�д�
������ʱ����ҵ����ϵ�д�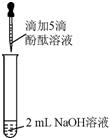 ������֪����̪��Һ��NaOH��Һ��죮���ǣ��ڷ���ʵ���У���ͼ��ʾ�������������벻����������Һ������ɫ��
������֪����̪��Һ��NaOH��Һ��죮���ǣ��ڷ���ʵ���У���ͼ��ʾ�������������벻����������Һ������ɫ����������⡿����ʵ���У��������벻���������ԭ����ʲô�أ�
����������衿
��ɫ��ȥ����NaOH��Һ�Ϳ����е�CO2��Ӧ�йأ�
��ɫ��ȥ����NaOH��Һ��Ũ���йأ�
����ʵ�顿
| ʵ�� | ʵ����� | ʵ������ |
| 1 | ��ʢ��2mLNa2CO3��Һ���Թ��еμ�5��0.5%�ķ�̪��Һ | ��Һ��� |
| 2 | 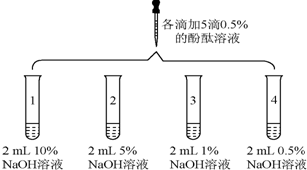 | 1���Թ�0.1min��ɫ��ȥ 2���Թ�5min��ɫ��ȥ 3���Թ�30min��ɫ���Ա�dz 4���Թ�120min��ɫ�����Ա仯 |
��1��ʵ��1��������������������1���������������������������
��2����ʵ��2�ó����ۣ����֡���Һ������ɫ�������ԭ����������������Һ��Ũ���йأ�����������ҺŨ�ȹ���ʹ��̪��ɫ��
| A�� | ̼���ƺ�̼��� | B�� | ����غ��Ȼ��� | C�� | ����ͭ���Ȼ��� | D�� | �����ƺ����ᱵ |
��1����һС������ϡ����ⶨ��Ʒ��Na2CO3�������������õ�����ƽ�����������±���
| �� �� �� Ŀ | �������ˣ� |
| ��ȡ��Ʒ | 9.30 |
| ��ƿ���� | 41.20 |
| ��ƿ+ϡ���������������� | 141.20 |
| ��ƿ+ϡ��������+ȫ����Ʒ��һ�γ��������� | 148.50 |
| ��ƿ+ϡ��������+ȫ����Ʒ�ڶ��γ��������� | 148.30 |
| ��ƿ+ϡ��������+ȫ����Ʒ�����γ��������� | 148.30 |
�ڻ������Na2CO3����������Ϊ57%��
��2���ڶ���ѡ����һ�ַ����ⶨ��Ʒ��ȡm�ˣ���Na2CO3������������������������£�
��������$\stackrel{ˮ}{��}$��Һ$��_{����B}^{A��Һ}$BaCO3$��_{����}^{ϴ�Ӹ���}$a��
��A��BaCl2���ѧʽ������ʵ��Ҫ������A��Һ������������A��Һ�Ѿ������ķ�������������Һ�еμ�̼������Һ���а�ɫ����������
�ڼ���������Na2CO3��������������д�����㲽�裮��
| A�� | 4.08g | B�� | 8.17g | C�� | 12.26g | D�� | 16.34g |
 ����ͼ��ʾ��װ���У�����ƿ�ڳ���������̼���壬ƿ�ڵ�˫����һ�ײ�һ����������IJ����ܣ���һ�ײ�һ֧ʢ������������Һ�Ľ�ͷ�ιܣ��ѽ�ͷ�ι��е�����������Һ����ƿ�У��������������ͣ������������ԭ������̼������������Һ��Ӧʹƿ��ѹǿ��С��
����ͼ��ʾ��װ���У�����ƿ�ڳ���������̼���壬ƿ�ڵ�˫����һ�ײ�һ����������IJ����ܣ���һ�ײ�һ֧ʢ������������Һ�Ľ�ͷ�ιܣ��ѽ�ͷ�ι��е�����������Һ����ƿ�У��������������ͣ������������ԭ������̼������������Һ��Ӧʹƿ��ѹǿ��С�� ����̼��þ ����ȡ����þ�����ϡ����ᡢ���û�ױƷ�Ĺ�ҵԭ�ϣ�
����̼��þ ����ȡ����þ�����ϡ����ᡢ���û�ױƷ�Ĺ�ҵԭ�ϣ�