��Ŀ����
ͬѧ�Ƕ�ʵ�顰����ʯ��ʯ��չ������̽����
������ʯ��ʯ���ɹ۲쵽ʯ��ʯ�ı���______��
��Ϊ֤��ʯ��ʯ�ѷֽ⣬��λͬѧ�������Ƕ���Ʒ������£�
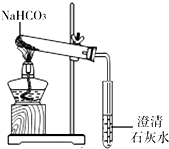
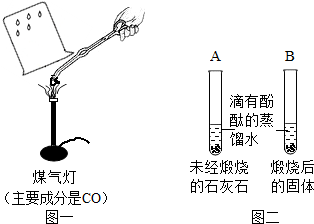
��ͬѧ��ͼһ��ʾ����ʵ�飨ú�������ܴﵽʯ��ʯ�ֽ���¶ȣ����۲쵽�ձ��ڱ�ʯ��ˮ����ǣ�д��ʯ��ˮ������Ӧ�Ļ�ѧ����ʽ______��
����ͬѧ��ͼ����ʾ�������飬�۲쵽B��Һ���______ɫ��A��ʵ�������______��
��ͬѧȡһ��ʯ��ʯ�������գ�һ��ʱ����ֹ�����������mg����CO2����������֤��ʯ��ʯ�ѷֽ⣮
IV�������Ϊ��ͬѧ�ķ����������������ԭ��______��
��Ϊ�ⶨʯ��ʯ�Ĵ��ȣ���ͬѧ�����ղ����պ�Ĺ����������ϡ�����У��ֲ���mg���壬����������ݼ����֪������ȡ�����ʯ��ʯ�к�̼���______mol����m��ʾ����
������ʯ��ʯ���ɹ۲쵽ʯ��ʯ�ı���______��
��Ϊ֤��ʯ��ʯ�ѷֽ⣬��λͬѧ�������Ƕ���Ʒ������£�
��ͬѧ��ͼһ��ʾ����ʵ�飨ú�������ܴﵽʯ��ʯ�ֽ���¶ȣ����۲쵽�ձ��ڱ�ʯ��ˮ����ǣ�д��ʯ��ˮ������Ӧ�Ļ�ѧ����ʽ______��
����ͬѧ��ͼ����ʾ�������飬�۲쵽B��Һ���______ɫ��A��ʵ�������______��
��ͬѧȡһ��ʯ��ʯ�������գ�һ��ʱ����ֹ�����������mg����CO2����������֤��ʯ��ʯ�ѷֽ⣮
IV�������Ϊ��ͬѧ�ķ����������������ԭ��______��
��Ϊ�ⶨʯ��ʯ�Ĵ��ȣ���ͬѧ�����ղ����պ�Ĺ����������ϡ�����У��ֲ���mg���壬����������ݼ����֪������ȡ�����ʯ��ʯ�к�̼���______mol����m��ʾ����

��ʯ��ʯ�ֽ�����������̼�����Կ��Կ���ʯ��ʯ�ı����������ݣ�
�ڢ�ʯ��ˮ����������ڶ�����̼�ͳ���ʯ��ˮ�����˷�Ӧ���÷�Ӧ������������ˮ��̼��ƣ��仯ѧ����ʽΪ��Ca��OH��2+CO2�TCaCO3��+H2O��
�������ƿ��Ժ�ˮ��Ӧ�����������ƣ�������������Һ�ʼ��ԣ�����ʹ��̪��Һ��죬���Կ��Թ۲쵽�Թ�B��Һ���죬�Թ�A���˶������ã�
����ú���Ƶ���Ҫ�ɷ�Ϊһ����̼����һ����̼ȼ�������˶�����̼�����Գ���ʯ��ˮ����Dz�һ������Դ��̼��Ƶķֽ⣻
�۸���������Ϣ����֪�����ɶ�����̼�����������Ϊ2m�ˣ�̼��ƵĻ�ѧʽ���Էֲ�Ϊ��CaO?CO2������̼��Ƶ����ʵ���Ϊ��2mg�£�
��100%����100g/mol=
mol
�ʴ�Ϊ���������ݲ�����
�ڢ�Ca��OH��2+CO2�TCaCO3��+H2O��
�죻���գ�
����ú���Ƶ���Ҫ�ɷ�Ϊһ����̼����һ����̼ȼ�������˶�����̼�����Գ���ʯ��ˮ����Dz�һ������Դ��̼��Ƶķֽ⣻
��
��
�ڢ�ʯ��ˮ����������ڶ�����̼�ͳ���ʯ��ˮ�����˷�Ӧ���÷�Ӧ������������ˮ��̼��ƣ��仯ѧ����ʽΪ��Ca��OH��2+CO2�TCaCO3��+H2O��
�������ƿ��Ժ�ˮ��Ӧ�����������ƣ�������������Һ�ʼ��ԣ�����ʹ��̪��Һ��죬���Կ��Թ۲쵽�Թ�B��Һ���죬�Թ�A���˶������ã�
����ú���Ƶ���Ҫ�ɷ�Ϊһ����̼����һ����̼ȼ�������˶�����̼�����Գ���ʯ��ˮ����Dz�һ������Դ��̼��Ƶķֽ⣻
�۸���������Ϣ����֪�����ɶ�����̼�����������Ϊ2m�ˣ�̼��ƵĻ�ѧʽ���Էֲ�Ϊ��CaO?CO2������̼��Ƶ����ʵ���Ϊ��2mg�£�
| 44 |
| 100 |
| m |
| 22 |
�ʴ�Ϊ���������ݲ�����
�ڢ�Ca��OH��2+CO2�TCaCO3��+H2O��
�죻���գ�
����ú���Ƶ���Ҫ�ɷ�Ϊһ����̼����һ����̼ȼ�������˶�����̼�����Գ���ʯ��ˮ����Dz�һ������Դ��̼��Ƶķֽ⣻
��
| m |
| 22 |

��ϰ��ϵ�д�
 һ����������ϵ�д�
һ����������ϵ�д�
�����Ŀ