��Ŀ����
11�� 2010��4��28��ijý����Ϊ�����۹���ʩ���������ŵ����ˡ��ı���������ij��ȤС��ͬѧ��˼����
2010��4��28��ijý����Ϊ�����۹���ʩ���������ŵ����ˡ��ı���������ij��ȤС��ͬѧ��˼������������⡿���۹ܵ��еĶ�����ʲô�ɷ֣�
���������ϡ�
I�����۹ܵ��еĴ��л�����һ�������·��ͻ����CO��CO2��H2S��CH4�ȣ�
��H2S��������CuSO4��Һ��Ӧ���ɺ�ɫ������
��������롿С��ͬѧ�����۹ܵ���������������������3�ֳɷֵIJ������£�
����1����CO��CO2��H2S�� ����2����CO��CO2��CH4�� ����3����CO��H2S��CH4��
����4����CO2��H2S��CH4�� ����5����CO��CO2��H2S��CH4��
��ʵ�鷽����С��ͬѧ��ͬ�������ͼ��ʾ��װ�ò�����̽�����г�������ʡ�ԣ���
���������ۡ�
��1�����Aװ��û�����Ա仯�������2���������Bװ��û�����Ա仯�������3������
��2������֤����l��ʵ���У�װ��C��NaOH��Һ������������CO2����Ҫ��һ����֤����ȼ�պ�IJ�������ǣ�Ѹ�ٰ��ձ������������ձ���ע�����ʯ��ˮ����
��3��Ҫȷ֤����5���Ƿ���CH4��ijͬѧ��Ϊͼʾװ���в���֮������Ҫ��װ��C��D֮���һ������װ�ã��Ľ���������ȼ�գ���Dװ���ڱڳ���ˮ����֤��������һ������CH4��Ϊ�˽�һ��ȷ���������Ƿ���
CO���ɷֱ�ⶨȼ�ղ�����H2O��CO2���������䷽���ǣ���ȼ�ղ�������ͨ��ʢ��ŨH2SO4��NaOH��Һ��װ�ã��ֱ��������ȼ�ղ���ǰ������ȼ�ղ����װ�õ�������ͨ�����㡢�����ó����ۣ�
���� �������CO�Ļ�ԭ�ԣ�CO2��ʹ����ʯ��ˮ����ǡ�CH4ȼ�����ɶ�����̼��ˮ�����ʣ������Ŀ�е���Ϣ�����۹ܵ��еĴ��л�����һ�������·��ͻ����CO��CO2��H2S��CH4�Ⱥ�H2S��������CuSO4��Һ��Ӧ���ɺ�ɫ�������ٹ۲�ͼ��ʵ�鲽������ͭ��Һ���������̼��B����ʯ��ˮ���������̼��C����������Һ���տ����еĶ�����̼�����ɿ��ٽ���⣮
��� �⣺��������롿�������۹ܵ��еĴ��л�����һ�������·��ͻ����CO��CO2��H2S��CH4����һ��Ϣ����������������IJ²⣬��֪����4������CH4��
���������ۡ���1��A��������ͭ��Һ���������ⷴӦ���ɺ�ɫ��������������֤���������⣬�����2������B���dz����ʯ��ˮ��������̼��ʹ������ǣ���Bװ�������ԣ�˵������������̼��
��2������֤����l��ʵ���У�Ҫ��֤һ����̼������һ����̼ȼ�����ɶ�����̼������ԭ���Ķ�����̼Ҫ������ȥ���������������մ����Ķ�����̼����3����֤�������ü���ȼ���ܹ�����ˮ�Ͷ�����̼���ʼ���ȼ��ǰ��ˮ�Ͷ�����̼�����ȥ��������C��D֮���һ������װ�ã����ɶ�����ձ�����ˮ��֤�����м��飮Ҫ��֤�Ƿ�һ����̼�����������ɵ�ˮ��Ȼ�������������������ɵĶ�����̼�����Ƴ�������װ��ʵ��ǰ�������ı仯��
�ʴ�Ϊ����������롿CH4�����������ۡ���1��2��3����2������CO2������ʯ��ˮ����3�����ˮ����ŨH2SO4����CaCl2����ˮCuSO4���� NaOH��Һ��
���� ������Ϣ��ʵ��̽���⣬ֻ�������������ʵ����ʲ���ȷ˳���ĵó���ȷ�𰸣�

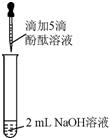 ������֪����̪��Һ��NaOH��Һ��죮���ǣ��ڷ���ʵ���У���ͼ��ʾ�������������벻����������Һ������ɫ��
������֪����̪��Һ��NaOH��Һ��죮���ǣ��ڷ���ʵ���У���ͼ��ʾ�������������벻����������Һ������ɫ����������⡿����ʵ���У��������벻���������ԭ����ʲô�أ�
����������衿
��ɫ��ȥ����NaOH��Һ�Ϳ����е�CO2��Ӧ�йأ�
��ɫ��ȥ����NaOH��Һ��Ũ���йأ�
����ʵ�顿
| ʵ�� | ʵ����� | ʵ������ |
| 1 | ��ʢ��2mLNa2CO3��Һ���Թ��еμ�5��0.5%�ķ�̪��Һ | ��Һ��� |
| 2 | 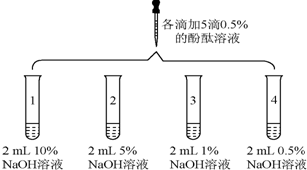 | 1���Թ�0.1min��ɫ��ȥ 2���Թ�5min��ɫ��ȥ 3���Թ�30min��ɫ���Ա�dz 4���Թ�120min��ɫ�����Ա仯 |
��1��ʵ��1��������������������1���������������������������
��2����ʵ��2�ó����ۣ����֡���Һ������ɫ�������ԭ����������������Һ��Ũ���йأ�����������ҺŨ�ȹ���ʹ��̪��ɫ��
��1����һС������ϡ����ⶨ��Ʒ��Na2CO3�������������õ�����ƽ�����������±���
| �� �� �� Ŀ | �������ˣ� |
| ��ȡ��Ʒ | 9.30 |
| ��ƿ���� | 41.20 |
| ��ƿ+ϡ���������������� | 141.20 |
| ��ƿ+ϡ��������+ȫ����Ʒ��һ�γ��������� | 148.50 |
| ��ƿ+ϡ��������+ȫ����Ʒ�ڶ��γ��������� | 148.30 |
| ��ƿ+ϡ��������+ȫ����Ʒ�����γ��������� | 148.30 |
�ڻ������Na2CO3����������Ϊ57%��
��2���ڶ���ѡ����һ�ַ����ⶨ��Ʒ��ȡm�ˣ���Na2CO3������������������������£�
��������$\stackrel{ˮ}{��}$��Һ$��_{����B}^{A��Һ}$BaCO3$��_{����}^{ϴ�Ӹ���}$a��
��A��BaCl2���ѧʽ������ʵ��Ҫ������A��Һ������������A��Һ�Ѿ������ķ�������������Һ�еμ�̼������Һ���а�ɫ����������
�ڼ���������Na2CO3��������������д�����㲽�裮��
��ȡ5����С��ͬ�ļ���ƿ���ֱ���1��2��3��4��5�����ֱ�ʹƿ������ռ����ƿ�ݻ�10%��20%��30%��40%��
50%��ˮ������Ϊ���������ò���Ƭ��ס��������װ��ˮ��ˮ���У�
�ڷֱ�����ˮ������5��ƿ����ͨ��������С�ĵذ�5��ƿ�ӵ�ˮ������ò���Ƭ��סƿ�ڣ�ȡ�����źã�
�۷ֱ����ǵ�ľ������5��ƿ�У��۲쵽���������£�
| ��� | 1 | 2 | 3 | 4 | 5 |
| ���� | �� | �� | ���� | ��ȼ | ��ȼ |
��1���ӹ۲�������֪��ʹ�����ǵ�ľ����ȼ������������С�����������3��ƿ��4��ƿ֮�䣮
��2������ˮ���ռ����������4ƿ���������������ԼΪ52.6%��
��3��ʹ�ô�����ľ���������������ķ����Ƿ�ɿ�������ǡ�����
��4����Ҫ�õ���ȷ�����������Χ��ֻ������ٲ����м���ƿ��ˮ����������ظ�����ʵ�鲽�輴�ɣ���ȡ����ֵ����̽���ܼ���ʵ�����������Ϊ������ʵ��Ӧ����ƿ��ˮռ����ƿ�ݻ����������Ϊ35%��
| A�� | 4.08g | B�� | 8.17g | C�� | 12.26g | D�� | 16.34g |
 ��ͼ��a��b��c�������ʵ��ܽ�����ߣ�a��c���ܽ�������ཻ��P�㣮��ͼ�ش�
��ͼ��a��b��c�������ʵ��ܽ�����ߣ�a��c���ܽ�������ཻ��P�㣮��ͼ�ش�
 �ס��ҡ����������ʵ��ܽ��������ͼ��ʾ����ͼ�ش�
�ס��ҡ����������ʵ��ܽ��������ͼ��ʾ����ͼ�ش�