��Ŀ����
2��þ���仯���������������������Ź㷺��Ӧ��ǰ������һ��þ�Ĺ㷺Ӧ��
��1��û��þ�Ĵ��ڣ��Ͳ����γ�Ҷ���أ�Ҳ��û�й�����ã������þ��ָB��
A������ B��Ԫ�� C��ԭ��
��2��������̥�ɲ����˿���ϳ�����ɣ�����þ����̥�Ĵٽ���������ǿ��Ӳ�ȣ�
���������в��漰��C��ѡ����ţ���
A���Ͻ� B���ϳɲ��� C�����ǽ������� D�����ϲ���
��3������þ������������������ȼ��ʱ����ҫ�۵�ǿ�⣮
������MgO���Ʊ�
��ҵ���ð���ʯ����MgCO3��CaCO3����ȡ��������þ��MgO������Ҫ������ͼ1��
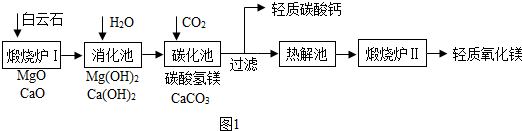
��1��д������¯���е�һ����ѧ��Ӧ����ʽCaCO3$\frac{\underline{\;����\;}}{\;}$CaO+CO2������MgCO3$\frac{\underline{\;����\;}}{\;}$MgO+CO2����
��2��̼���ص������ǽ���Ԫ��ת��Ϊ��������ѧ��Ӧ����ʽ��Ca��OH ��2+CO2=CaCO3��+H2O��
��3���������л��γɷ��ڵ�����Һ�����ڵ�ԭ���������������ˮ��Ӧ���ȣ�
��4��̼����þ����ɺ�������С�մ����ƣ����Ƚ������Һ�����ʻ�ѧʽ��Mg��HCO3��2��
��������ʽ̼��þ��ɲⶨ
����¯���������Ǽ�ʽ̼��þ��aMgCO3•bMg��OH��2•cH2O����Ϊȷ����ɷ֣�����ͼ2ʵ�飨����ÿ����Ӧ�����ն���ȫ����
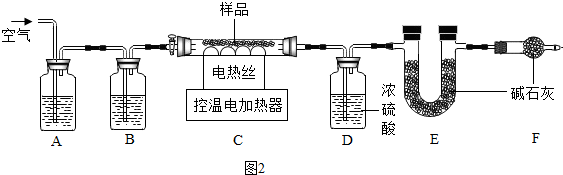
���������ϡ���ʽ̼��þ���ȷֽ�����MgO��CO2��H2O��
��1��װ���ʵ��װ�ú�����Ҫ���װ�������ԣ�
��2��A��Bװ�õ������dz�ȥ�����еĶ�����̼��ˮ������ϴ��ƿB��Ӧʢ��a��ѡ����ţ���
a��Ũ���� b�������ʯ��ˮ c������������Һ
��3���ֳ�ȡ48.4g��ʽ̼��þ��Cװ���н����ȷֽ⣬ֱ��Cװ����ʣ������������ٱ仯Ϊֹ������ͨ��������ȴ����ȴ��װ��D����10.8g��װ��E����17.6g������ͨһ��ʱ�������Ŀ����ʹ��Ӧ���ɵ�ˮ�Ͷ�����̼����ȫ���գ�
��4�������������ݣ�������MgO������20g��a?b?c=4?1?5��
��5����ȱ��Fװ�ã���õ�aֵƫ��ѡ�ƫ����ƫС���������䡱����
���� ��һ��þ�Ĺ㷺Ӧ��
þԪ���ǽ��й�����õı���Ԫ�أ�
���ϰ����������ϣ����������ͺϽ𣩡����ǽ������ϡ����ϲ��ϵȣ�
þ�ڿ�����ȼ��ʱ����ҫ�۵�ǿ�⣬��������þ��
������MgO���Ʊ�
���������£�̼��Ʒֽ����������ƺͶ�����̼��̼��þ�ֽ���������þ�Ͷ�����̼��
�������ƺͶ�����̼��Ӧ���ɰ�ɫ����̼��ƺ�ˮ��
�����ƺ�ˮ��Ӧ�����������ƣ�ͬʱ�ų��������ȣ�
��������ʽ̼��þ��ɲⶨ
����������μӻ������ʵ�飬ʵ��ǰһ��Ҫ���װ�õ������ԣ��Է�װ��©��Ӱ��ʵ������
����������Һ�ܹ����ն�����̼���壬Ũ�����ܹ�����ˮ������
��ʯ���ܹ�����ˮ�����Ͷ�����̼��
��� �⣺��һ��þ�Ĺ㷺Ӧ��
��1��û��þ�Ĵ��ڣ��Ͳ����γ�Ҷ���أ�Ҳ��û�й�����ã������þ��ָԪ�أ�
���B��
��2�������˿���ںϽ𣬺ϳ������ںϳɲ��ϣ�������̥���ڸ��ϲ��ϣ����漰�����ǽ������ϣ�
���C��
��3������þ������������������ȼ��ʱ����ҫ�۵�ǿ�⣮
���ҫ�۵�ǿ�⣮
������MgO���Ʊ�
��1�����������£�̼��Ʒֽ����������ƺͶ�����̼��̼��þ�ֽ���������þ�Ͷ�����̼����Ӧ�Ļ�ѧ����ʽΪ��CaCO3$\frac{\underline{\;����\;}}{\;}$CaO+CO2����MgCO3$\frac{\underline{\;����\;}}{\;}$MgO+CO2����
���CaCO3$\frac{\underline{\;����\;}}{\;}$CaO+CO2������MgCO3$\frac{\underline{\;����\;}}{\;}$MgO+CO2����
��2��̼���ص������ǽ���Ԫ��ת��Ϊ��������ѧ��Ӧ����ʽ�ǣ�Ca��OH ��2+CO2=CaCO3��+H2O��
���Ca��OH ��2+CO2=CaCO3��+H2O��
��3���������л��γɷ��ڵ�����Һ�����ڵ�ԭ���������������ˮ��Ӧ���ȣ�
�����������ˮ��Ӧ���ȣ�
��4��̼����þ����ɺ�������С�մ����ƣ����Ƚ������Һ��������̼����þ��̼����þ��ѧʽ��Mg��HCO3��2��
���Mg��HCO3��2��
��������ʽ̼��þ��ɲⶨ
��1��װ���ʵ��װ�ú�����Ҫ���װ�������ԣ�
������װ�������ԣ�
��2��A��Bװ�õ������dz�ȥ�����еĶ�����̼��ˮ������
ϴ��ƿB��Ӧʢ��Ũ���ᣮ
�����ȥ�����еĶ�����̼��ˮ������a��
��3������ͨһ��ʱ�������Ŀ����ʹ��Ӧ���ɵ�ˮ�Ͷ�����̼����ȫ���գ�
���ʹ��Ӧ���ɵ�ˮ�Ͷ�����̼����ȫ���գ�
��4�������������ݿ�֪�����ɶ�����̼������17.6g������ˮ��������10.8g��������MgO�����ǣ�48.4g-10.8g-17.6g=20g��
��̼��þ����Ϊx����������þ����Ϊy��
MgCO3$\frac{\underline{\;����\;}}{\;}$MgO+CO2����
84 40 44
x y 17.6g
$\frac{84}{x}$=$\frac{40}{y}$=$\frac{44}{17.6g}$��
x=33.6g��y=16g��
������þ�ֽ���������þ����Ϊ��20g-16g=4g��
��������þ����Ϊm������ˮ������Ϊn��
Mg��OH��2$\frac{\underline{\;����\;}}{\;}$MgO+H2O��
58 40 18
m 4g n
$\frac{58}{m}$=$\frac{40}{4g}$=$\frac{18}{n}$��
m=5.8g��n=1.8g��
ˮ������Ϊ��10.8g-1.8g=9g��
����������$\frac{84a}{33.6g}$=$\frac{58b}{5.8g}$=$\frac{18c}{9g}$��
a?b?c=4?1?5��
���20��4?1?5��
��5����ȱ��Fװ�ã������е�ˮ�����Ͷ�����̼�����Eװ���У����²�õ�aֵƫ��
���ƫ��
���� �������ʵ�飬��ѧ�ؽ���ʵ�顢����ʵ�飬�ǵó���ȷʵ����۵�ǰ�ᣬ���Ҫѧ�����ʵ�顢����ʵ�顢����ʵ�飬Ϊѧ�û�ѧ֪ʶ�춨������

| A�� | ��ũ�ҷ��뻯���ۺ�ʹ�ã����������Ч�� | |
| B�� | �Դ����е�ֲ��ʩ��������CO2���Դٽ�������� | |
| C�� | ��ֲ����ֳ�����������ϣ��ȿ��Ը��ƻ����ֿ������ũ����ҵ���� | |
| D�� | ���Ȼ�狀���ʯ�һ��ʹ�ã��ڸ�ũ�����ṩӪ��Ԫ��ͬʱ���ܽ������� ���� |
| A�� | ��ֲ��ĺ����벻���� | B�� | ��������ˮ | ||
| C�� | ���������ԣ���һ�ֳ��õ������� | D�� | �ܹ�ȼ�� |
| A�� | X2Y | B�� | XY2 | C�� | X2Y2 | D�� | X3Y2 |
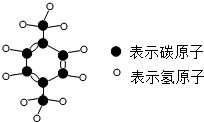 PX����ɫ��Һ�壬���з�����ζ�������Ҵ����л��ܼ����ܣ���ȼ���Ͷ��������Ը����Ҵ�����������������γɱ�ը�Ի�����ͼ�ǡ�PX���Ľṹģ�ͣ������йء�PX����˵������ȷ���ǣ�������
PX����ɫ��Һ�壬���з�����ζ�������Ҵ����л��ܼ����ܣ���ȼ���Ͷ��������Ը����Ҵ�����������������γɱ�ը�Ի�����ͼ�ǡ�PX���Ľṹģ�ͣ������йء�PX����˵������ȷ���ǣ�������| A�� | ��PX�������л������� | |
| B�� | ��PX����ȼ���Ͷ����������Ļ�ѧ���� | |
| C�� | ��PX���к���8��̼ԭ�ӡ�10����ԭ�� | |
| D�� | ��PX����C��H����Ԫ�ص�������Ϊ48��5 |
 ��ͼ��ʾ����������ʾ��һ������ת��Ϊ��һ�����ʣ���--����ʾ�������ʼ���Է�Ӧ����������������ȥ��A--F���dz��л�ѧ�г��������ʣ�����A�dz������������ϣ�D������θҺ�е�һ�ֳɷ֣�E��F������������жϣ�
��ͼ��ʾ����������ʾ��һ������ת��Ϊ��һ�����ʣ���--����ʾ�������ʼ���Է�Ӧ����������������ȥ��A--F���dz��л�ѧ�г��������ʣ�����A�dz������������ϣ�D������θҺ�е�һ�ֳɷ֣�E��F������������жϣ�