��Ŀ����
ˮ�����ǵ������������еĹ�ϵ��
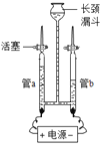
��1��Ϊ��֤ˮ�����������ͼ��ʾװ�ý���ʵ�顣
��д���÷�Ӧ�Ļ�ѧ����ʽ___________________��
�ڵ��ˮ�Ĺ����У������ı������_______��д���ƣ���
��������������ˮ���ܵ������______������ĸ����
A������ B���Ȼ��� C���Ȼ��� D����������
��2������200g��������Ϊ5%������������Һ����Ҫ��Ҫˮ____mL������ʱ�õ�����������________���ձ����������ͽ�ͷ�ιܡ���ˮ���ܶȽ��ƿ���1g/cm3��
��3�����������������Һ��ij������Һ��Ӧ����ͼ���������ֻ��������õ�����ҺpH�仯ͼ��
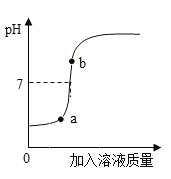
�ٸ���ͼ�����ߣ��жϽ��еIJ�����______������ĸ����
A����������Һ��εμӵ�����������Һ��
B��������������Һ��εμӵ�������Һ��
�ڴ��۽Ƕȷ������÷�Ӧʵ��Ϊ___________ ��
��b���Ӧ����Һ�е�����Ϊ____________д��ѧʽ)��
����80g 5%������������Һ��ǡ���к�50g������Һ���Լ����������Һ��������������_________��
 ״Ԫ��ȫ��ͻ�Ƶ�����ϵ�д�
״Ԫ��ȫ��ͻ�Ƶ�����ϵ�д� ֱͨ������У�ܲ��¿�ֱͨ��Уϵ�д�
ֱͨ������У�ܲ��¿�ֱͨ��Уϵ�д��ס��ҡ��������������ʵ����Ӧ��ϵ��ͼ��ʾ����������ʾ���������ʼ��ܷ�����Ӧ�����з��϶�Ӧ��Ӧ��ϵ��ѡ����

ѡ�� | A | B | C | D | |
�� �� | �� | Ca(OH)2 | CO2 | H2 | Mg |
�� | Na2CO3 | NaOH | O2 | HCl | |
�� | H2SO4 | CaCl2 | CuO | CuSO4 | |
�� | BaCl2 | K2CO3 | HCl | NaOH |
A. A B. B C. C D. D


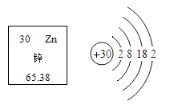
 X��CO2��������X�Ļ�ѧʽ��
X��CO2��������X�Ļ�ѧʽ��