��Ŀ����
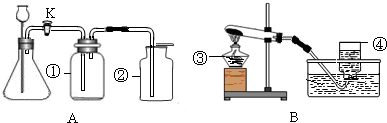
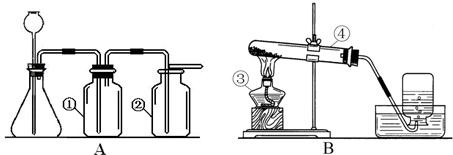
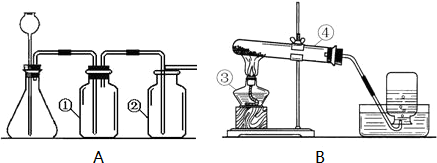
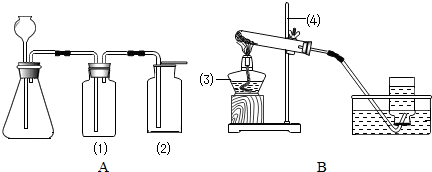
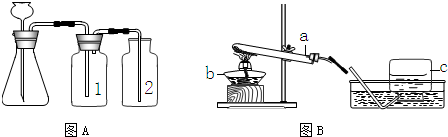
ʵ���ҳ�����ͼA��Bװ����ȡ���壬�ش�������⣺
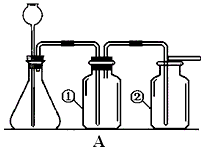

��1��д��ͼ�б��Тۡ��ܵ��������ƣ���____________����____________��
��2��С��Ҫ��ȡ������̼���壬Ӧѡ��ͼ�е�_____ _װ�ã�����ĸ����ʹ�õ�ҩƷ��__________��_________��д���÷�Ӧ�Ļ�ѧ����ʽ________________________________��
��3��С��Ҫ��Aװ����ȡ��������壬��ƿ��Ӧʢ��_______������ţ���
A������������Һ B��Ũ���� C��Ũ����
��4��д����Bװ����ȡ�����һ����ѧ��Ӧ����ʽ_________________________________��
��5��Aװ�������Եļ��飺�ֱ��ˮ��û����ƿ���е��ܵ�ĩ�˺���ƿ�г���©����ĩ�ˣ�Ȼ������ë����סϴ��ƿ�٣�������ƿ����______________________________����ƿ��________________________�����װ��©����
��2��С��Ҫ��ȡ������̼���壬Ӧѡ��ͼ�е�_____ _װ�ã�����ĸ����ʹ�õ�ҩƷ��__________��_________��д���÷�Ӧ�Ļ�ѧ����ʽ________________________________��
��3��С��Ҫ��Aװ����ȡ��������壬��ƿ��Ӧʢ��_______������ţ���
A������������Һ B��Ũ���� C��Ũ����
��4��д����Bװ����ȡ�����һ����ѧ��Ӧ����ʽ_________________________________��
��5��Aװ�������Եļ��飺�ֱ��ˮ��û����ƿ���е��ܵ�ĩ�˺���ƿ�г���©����ĩ�ˣ�Ȼ������ë����סϴ��ƿ�٣�������ƿ����______________________________����ƿ��________________________�����װ��©����
��1���۾ƾ��ƣ����Թ�
��2��A��CaCO3��ʯ��ʯ�����ʯ�������CaCO3��2HCl��CaCl2��H2O��CO2��
��3��C
��4��2KMnO4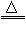 K2MnO4+MnO2+O2��
K2MnO4+MnO2+O2��
��5��û������ð����Һ�治�½�������©��ĩ��ˮ��������
��2��A��CaCO3��ʯ��ʯ�����ʯ�������CaCO3��2HCl��CaCl2��H2O��CO2��
��3��C
��4��2KMnO4
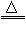 K2MnO4+MnO2+O2��
K2MnO4+MnO2+O2�� ��5��û������ð����Һ�治�½�������©��ĩ��ˮ��������

��ϰ��ϵ�д�
 ��ѧ����ϵ�д�
��ѧ����ϵ�д�
�����Ŀ