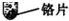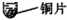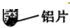��Ŀ����
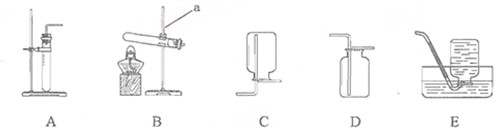
��11�֣�ʵ������ȡ�������������װ������ͼ��ʾ��

��ش��������⡣
��1����������������a_______��b_______��
��2����п����ϡ������ȡ�����Ļ�ѧ����ʽΪ_______���ø��������ȡ����ʱ����ѡ�õķ���װ��Ϊ____������ĸ��ţ���������Ӧ�Ļ�ѧ����ʽ�ǣ�_____��
��3���ô���ʯ��ϡ������ȡ������̼�Ļ�ѧ����ʽΪ_______����װ��_______������ĸ��ţ��ռ�������̼����ȼ�ŵ�ľ�����ڼ���ƿ�ڣ����۲쵽_______�� ˵��ƿ���ѳ���������̼��
���𰸡�
��1��a����ƿ b����©��
��2��Zn+2HCl=ZnCl2+H2��
A 2KMnO4  K2MnO4
+ MnO2 + O2��
K2MnO4
+ MnO2 + O2��
��3��CaCO3+2HCl=CaCl2+H2O+CO2�� C ����Ϩ��
��������
���������
�Ƹ����������������Ӧ����ȣ�ѡ���оƾ��Ƶ�Aװ�á�
�Ƕ�����̼�ܶȱȿ�������������ˮ������ֻ���������ſ������ռ���
���㣺�������ȡ������

��ϰ��ϵ�д�
 ��һ������ĩ�ٷֳ�̾�ϵ�д�
��һ������ĩ�ٷֳ�̾�ϵ�д�
�����Ŀ