��Ŀ����
12�� ˮ��һ����Ҫ����Դ��
ˮ��һ����Ҫ����Դ����1�����ˮʵ���ʾ��ˮ����ɣ���ʵ����ˮ�м����������Ƶ�Ŀ������ǿˮ�ĵ����ԣ���ͼ1ʵ���Թ�1���ռ�����������H2����ʵ����������������֧�Թ��ռ�������������ԼΪ1��2��д���÷�Ӧ�Ļ�ѧ����ʽ2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2�������ʵ��˵��ˮ������Ԫ�ء���Ԫ����ɵģ�
��2������ٳ�һ�������н�Լ��ˮ�Ĵ�ʩ����ˮ����������һ�����ɣ�
��3����ˮ�����ɻ��ˮ��Դ�ѷ������⣮ͼ2Ϊ̫���ܺ�ˮ����װ��ʾ��ͼ��
��ˮ���ˮ�����Ĺ����У��������仯����AB������ĸ��ţ���
A���������� B���������� C�����Ӽ����
���� ��1���������е�֪ʶ���з��������ˮʱ�������ɵ����������������ɵ����������������������������Ϊ2��1��
��2�����ݽ�Լ��ˮ�Ĵ�ʩ���н��
��3������ˮ���ˮ�����Ĺ������������仯���
��� �⣺��1�����ˮʵ���ʾ��ˮ����ɣ���ʵ����ˮ�м����������Ƶ�Ŀ������ǿˮ�ĵ����ԣ���ͼ1ʵ���Թ�1���Դ�ĸ��������������Թ����ռ�����������������
�������ɵ�����������ʵ����������������֧�Թ��ռ�������������ԼΪ1��2��ˮͨ��ֽ������������������÷�Ӧ�Ļ�ѧ����ʽ2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2�������ʵ��˵��ˮ������Ԫ�ء���Ԫ����ɵģ�
��2�������н�Լ��ˮ�Ĵ�ʩ��һˮ���ã���������ˮ������
��3��ˮ���ˮ�����Ĺ������������仯�����������ͷ�������䣬���Ӽ���ı䣮
�ʴ�Ϊ����1����ǿˮ�ĵ����ԣ� H2��1��2��2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2���� ��Ԫ�ء���Ԫ�أ�
��2������ˮ������
��3��AB��
���� �����Ĺؼ���Ҫ���������������ֱ仯�������Ƿ����仯��ֻ�������˷��ӵ����ʲ��ܶ�����������ȷ���жϣ�

| A�� | ��������ƽ��ȡ12.62g��ʳ�� | |
| B�� | ��10ml����Ͳ��ȡ8.76ml��ˮ | |
| C�� | ��ͨ�¶ȼ�����ʾ�����¶���Ϊ25.67�� | |
| D�� | ��10ml��Ͳ��ȡij��Һ�����Ϊ8.2ml |
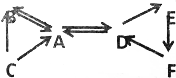 A-F��ʾ���л�ѧ���������ʣ�����֮���ת����ϵ��ͼ��ʾ����ͨ������£�A��B�����Ԫ����ͬ���������壬D��E��F���������ж����и�Ԫ�أ���DΪ����ʯ����Ҫ�ɷ֣���ش��������⣺
A-F��ʾ���л�ѧ���������ʣ�����֮���ת����ϵ��ͼ��ʾ����ͨ������£�A��B�����Ԫ����ͬ���������壬D��E��F���������ж����и�Ԫ�أ���DΪ����ʯ����Ҫ�ɷ֣���ش��������⣺