��Ŀ����
��ľ����ũ�ҷ��ϣ�������Ҫ�ɷ���̼��أ���������أ��Ȼ��صȣ���ѧ��ȤС��Ϊ�ⶨij��ľ����Ʒ�е���Ч�ɷ֣�ȡ100��ľ�����ձ��У����ϵ���ϡ������Һ��������30gϡ����ʱ�����������ݲ�������ʱ�ձ��еIJ������������Ϊ127.8g��
̼��������ᷴӦ�Ļ�ѧ����ʽΪK2CO3+H2SO4═K2SO4+CO2��+H2O
�������ľ�ҵ������ɷֲ�����Ԫ�أ������ᷴӦ��
�����ش�
��1��������̼��CO2�������У�̼����Ԫ�ص�ԭ�Ӹ�����Ϊ����
��2��̼��أ�K2CO3������Է�������Ϊ����
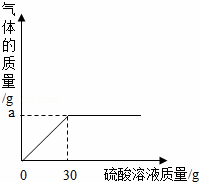
��3����ͼ��ʾ��Ӧ���̷ų��������������������Ĺ�ϵ���ߣ�����������غ㶨�����ͼ����������a����ֵ��
a=����g��
��4����ľ����Ʒ��̼��ص�������Ҫ��д��������̣�
��5��ͨ������ʵ�飬��ø�100g��ľ�������������Ϊ8.7g���Ȼ�������Ϊ2.98g����֪�طʵ���Ч�ɷ����Ȼ��ؼƣ���ò�ľ����Ʒ���Ȼ��ص���������Ϊ��2.98��%
| ��1�����ݻ�ѧʽ����ؼ���������������̼��̼ԭ�Ӻ���ԭ�ӵĸ����ȣ� ��2�����ݻ�ѧʽ�ļ�����Լ����̼��ص���Է��������� ��3�����������غ㶨�ɿ��Լ������������������� ��4���������������������Ϸ�Ӧ�Ļ�ѧ����ʽ���Լ����̼��ص������� ��5��������������������ɽ�ɣ� | |
| ��� | �⣺��1��������̼��CO2�������У�̼����Ԫ�ص�ԭ�Ӹ�����Ϊ1��2�� ��2��̼��أ�K2CO3������Է�������Ϊ��39��2+12+16��3=138�� ��3���������غ㶨�ɿ���֪������Ӧǰ����ٵ�������Ϊ���ɵĶ�����̼�����������Կ������ͼ����������a����ֵΪ��100g+30﹣127.8g=2.2g�� ��4����ľ����Ʒ��̼��ص�����Ϊx K2CO3+H2SO4═K2SO4+CO2��+H2O 138 44 x 2.2g
��ã�x=6.9g �𣺲�ľ����Ʒ��̼��ص�����Ϊ6.9g�� �ò�ľ����Ʒ���Ȼ��ص���������Ϊ�� �ʴ�Ϊ����1��1��2�� ��2��138�� ��3��2.2�� ��4��6.9g ��5��2.98�� |
��

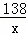 =
=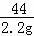
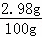 ��100%=2.98%��
��100%=2.98%��
 ��2013?���ݣ���ľ����ũ�ҷ��ϣ�������Ҫ�ɷ���̼��أ���������أ��Ȼ��صȣ���ѧ��ȤС��Ϊ�ⶨij��ľ����Ʒ�е���Ч�ɷ֣�ȡ100��ľ�����ձ��У����ϵ���ϡ������Һ��������30gϡ����ʱ�����������ݲ�������ʱ�ձ��еIJ������������Ϊ127.8g��
��2013?���ݣ���ľ����ũ�ҷ��ϣ�������Ҫ�ɷ���̼��أ���������أ��Ȼ��صȣ���ѧ��ȤС��Ϊ�ⶨij��ľ����Ʒ�е���Ч�ɷ֣�ȡ100��ľ�����ձ��У����ϵ���ϡ������Һ��������30gϡ����ʱ�����������ݲ�������ʱ�ձ��еIJ������������Ϊ127.8g��