��Ŀ����
5����ȼ���ױ���ѧҩƷ�Ĺ��������ѧ���淶���������ǵ������Ʋ��������ɹ�������ʧ��������ȥ������ۡ�8•12���ش���ֱ�ը�¹ʣ�������Ϊ����Σ�ջ�ѧҩƷ������淋�����������ȼ��������ƣ��Ĵ�Ź����������������˸������صĶ����ֺ�����֪����230�����ϣ�ͬʱ������Ӱ��ʱ����立ֽ����������2NH4NO3$\frac{\underline{\;\;��\;\;}}{\;}$2X��+O2��+4H2O������������ˮ���ҷ�Ӧ������������������ƣ�ͬʱ�ͷŴ������ȣ�����������Ϣ�ش��������⣺��1���ڱ������У���������ȷֽ�Ļ�ѧ����ʽ�У�X�Ļ�ѧʽΪN2��
��2����д�������ƺ�ˮ��Ӧ�Ļ�ѧ����ʽ2Na+2H2O=2NaOH+H2����
���� ��1�����������غ㶨�ɿ�֪���ڻ�ѧ��Ӧ�У���Ӧǰ��ԭ�ӵ�����û�иı䣬��Ŀû��������ԭ�ӵ�����Ҳû�иı䣻
��2������ˮ��Ӧ�����������ƺ�������
��� �⣺��1�����������غ㶨�ɺͻ�ѧ����ʽ��֪��X�Ļ�ѧʽ�к��е�Ԫ�ص�ԭ�Ӹ���Ϊ��4��2=2��������Ԫ�ص�ԭ�Ӹ���Ϊ����8-8��=0��������Ԫ�ص�ԭ�Ӹ���Ϊ��6-2-4=0����X�Ļ�ѧʽΪ��N2�����N2��
��2������ˮ��Ӧ�����������ƺ���������ѧ����ʽΪ��2Na+2H2O=2NaOH+H2�������2Na+2H2O=2NaOH+H2����
���� ������Ҫ����ѧ�����û�ѧ����ʽ�������غ㶨���Լ���ѧʽ�ۺϷ����ͽ��ʵ�������������������ѧ�����������˼ά��ȣ�ǿ����ѧ������֪ʶ��������

��ϰ��ϵ�д�
�����Ŀ
6��ͭƬ��Ũ������Է������·�Ӧ��Cu+4HNO3=Cu��NO3��2+2X��+2H2O�Ը÷�Ӧ������˵������ȷ���ǣ�������
| A�� | ������X�Ļ�ѧʽΪH2 | |
| B�� | ��Ӧ��NԪ�ػ��ϼ۸ı��ԭ����NԪ�ػ��ϼ۲����ԭ�ӵĸ�����Ϊ1��1 | |
| C�� | Ũ�������ڴ����� | |
| D�� | ��Ӧ��Cu��������H2O��������Ϊ32��9 |
13��ij��ѧС���ͬѧ̽��̼���Ʋ������ʣ�ʵ�鷽�������
����չ��Ӧ�á�Ϊ�˲ⶨʵ������һƿδ�Ǻ�ƿ�����ռ��Ƿ���ʼ����ʳ̶ȣ���С��ͬѧ����������ʵ�飺

��1������һ��������ɫ���������������Һ��죬���ۣ�����������Ʒ���ֱ���
��2��ʵ�鷽��������A��Һ������CD
A��ϡ���� B���������� C���Ȼ��� D������ƣ�
| ʵ��װ�� | ʵ�鲽�� | ʵ������ͽ��� |
 | ��1����ע�����е�X��ҺΪϡ���ᣬ����ע�������� ��2����ע�����е�X��Һע�������� | ��1���������ݣ���ɫ��Һ����ʧ ��2�������ְ�ɫ�����ͺ�ɫ����ʧ����X������Ca��OH��2��Ba��OH��2���ѧʽ�� |

��1������һ��������ɫ���������������Һ��죬���ۣ�����������Ʒ���ֱ���
��2��ʵ�鷽��������A��Һ������CD
A��ϡ���� B���������� C���Ȼ��� D������ƣ�
17������ʵ���������ȷ���ǣ�������
| A�� | 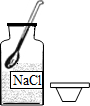 ȡ�ù���ҩƷ | B�� | 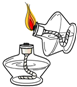 ��ȼ�ƾ��� | ||
| C�� | 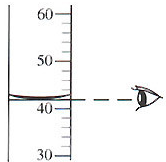 ��ȡҺ��ҩƷ | D�� | 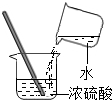 ϡ��Ũ���� |
