��Ŀ����
2009 �� 12 ��14�գ����о����˳��п�ѧ֪ʶӦ����̽�����ܱ��������е�Ҷ����ǩ������Ŀ����ѡ���ڹ涨ʱ���ڣ��������ѵĴ�����ƣ���Ҷ������Ⱦɫ���滭���øƴ�ӵ������ӹ�������ʱij��ѡ�������� 200�� 10%������������Һ���������������Ƕ�ʣ����������Ʒ�Һ������������кʹ������� 1 ������������Һʱ��Ҫ�������ƹ���______ �ˣ�
�� 2 ����ȫ�кͷ�Һʱ��ȥ 14.6%������ 100 �ˣ���ʣ���Һ�к������������ʶ��ٿˣ��������Һ�е��������ʾ��������ᷢ����Ӧ��

���𰸡��������������е�֪ʶ���з�������Һ�����ʵ�����=��Һ������×���ʵ����������������������ƺ����ᷴӦ�Ļ�ѧ����ʽ���н�ɣ�
����⣺��1��200g10%������������Һ���������Ƶ�����Ϊ��200g×10%=20g�����20��
��2����ʣ���Һ�к��������Ƶ�����Ϊx
NaOH+HCl=NaCl+H2O
40 36.5
x 100g×14.6%
 x=16g
x=16g
��ʣ���Һ�к�������������Ϊ16g��
���������⿼���˸��ݻ�ѧ����ʽ�ļ��㣬��ɴ��⣬�ؼ�������Ӧ��ԭ�����ҳ�������֮���������ϵȻ���б���ʽ���㣮
����⣺��1��200g10%������������Һ���������Ƶ�����Ϊ��200g×10%=20g�����20��
��2����ʣ���Һ�к��������Ƶ�����Ϊx
NaOH+HCl=NaCl+H2O
40 36.5
x 100g×14.6%
 x=16g
x=16g��ʣ���Һ�к�������������Ϊ16g��
���������⿼���˸��ݻ�ѧ����ʽ�ļ��㣬��ɴ��⣬�ؼ�������Ӧ��ԭ�����ҳ�������֮���������ϵȻ���б���ʽ���㣮

��ϰ��ϵ�д�
�����Ŀ
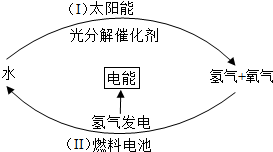 2009��12�£��籾���������������ٿ�����CO2Ϊ�������������ŷ��ٴγ�Ϊ������ע�Ľ��㣮
2009��12�£��籾���������������ٿ�����CO2Ϊ�������������ŷ��ٴγ�Ϊ������ע�Ľ��㣮