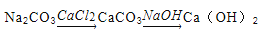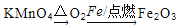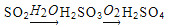��Ŀ����
������ҩ�������Ļ�ѧʽΪC47H51NO4������˵����ȷ����
A. �����������л��߷��ӻ����� B. ��������C��H��O��N����Ԫ�����
C. �������е�����Ԫ�ص�������Ϊ1��14 D. ����������Ԫ�ص������������
B ��������A. �����������л���������Ǹ߷��ӻ��������B. ��������C��H��O��N����Ԫ����ɣ���ȷ��C. �������е�����Ԫ�ص�������Ϊ14����16��4��=7:32������D. ��������̼Ԫ�ص��������������ѡB��
��ϰ��ϵ�д�
�����Ŀ