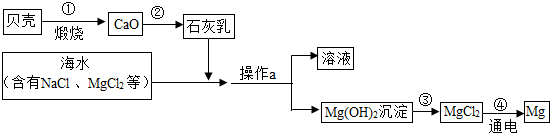
��Ŀ����
9����ѧ�������ǵ����ߣ��������ǵ�����ϢϢ��أ���ͼ��ijУ����ͬѧ�ռ���һЩ��־����ǩ����װ��ʵ��ʾ��ͼ����ͼ��ʾ����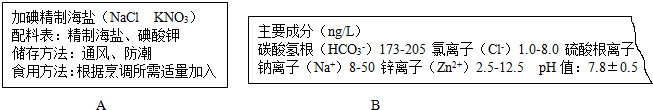
��1��ͼA��ij��ʳ�εİ�װ�����ӵ⺣���еġ��⡱��ָ��Ԫ�أ��ԭ�ӡ��������ӡ��������ӡ���Ԫ�ء��������û�ѧ������ȷ��ʾ������е�Ԫ�صĻ��ϼ�K$\stackrel{+5}{N}$O3��
��2��ͼB��ijƷ�ƿ�Ȫˮ�ı�ǩ������ֻ����������ӷ��ű���٣���������ӵķ���Ӧ����SO42-��������̪��Һ����ÿ�Ȫˮ��Ʒ�У����Ժ�ɫ������Ȼ��Ȫˮ�к��ж������ʣ�������һ�����ʿ���������θ�����֢���û�ѧ����ʽ��ʾ�䷴Ӧԭ��NaHCO3+HCl=NaCl+H2O+CO2����
���� ��1�����ʵ���ɳ���Ԫ�������������ݻ��������������ϼ�Ϊ0��ԭ�����Ԫ�صĻ��ϼۣ�
��2���������ӷ��ŵ�д��д�����ӵķ��ţ�̼�����ƿ���θҺ�й�������ᷴӦ������������θ����࣮
��� �⣺��1�����ʵ���ɳ���Ԫ�����������ӵ⺣���еġ��⡱��ָ��Ԫ�أ�������е�Ԫ�صĻ��ϼ�Ϊ+5�ۣ�������صĻ�ѧʽ�е�Ԫ�ط��ŵ����Ϸ������+5��������Ϊ��K$\stackrel{+5}{N}$O3�����Ԫ�أ�K$\stackrel{+5}{N}$O3��
��2����������ӵķ���Ӧ��������������ŵ����ϽDZ����2-������Ϊ��SO42-�����ڸÿ�Ȫˮ��pH����7���������ԣ�������̪��Һ����ÿ�Ȫˮ��Ʒ�У����Ժ�ɫ������Ȼ��Ȫˮ�к��ж������ʣ�������һ������̼�����ƿ���������θ�����֢���䷴Ӧԭ���ǣ�NaHCO3+HCl=NaCl+H2O+CO2�������SO42-���죻NaHCO3+HCl=NaCl+H2O+CO2����
���� �����ѶȲ�����Ҫ����ͬѧ�ǶԳ�����ѧ���ԭ�ӷ��š���ѧʽ�����ӷ��š����ϼ۵ȣ�����д������������

��ϰ��ϵ�д�
�����Ŀ
6�����������������ǣ�������
| A�� | ��ú¯�ķ����һ��ˮ��ú���ж� | |
| B�� | �÷�ù�Ļ���եʳ���� | |
| C�� | ʹ�ÿɽ������Ͻ������ɫ��Ⱦ������ | |
| D�� | �������Ѹ�ٴ��Ŵ�ͨ�� |
20��С��������100g10%��NaOH��Һ��������Ҷ����ǩ������������Һ�Ĺ����У�����������ȷ���ǣ�������
| A�� | �������ϵ�ֽ������10gNaOH���� | |
| B�� | ����ʱ����ָ������ƫת��Ӧ������������ʹ��ƽƽ�� | |
| C�� | ��100mL����Ͳ��ȡ90mL��ˮ | |
| D�� | ��NaOH���嵹��װ��90mLˮ����Ͳ���ܽ� |
4����������ѧ��ѧ֪ʶ�жϣ�������������ȷ���ǣ�������
| A�� | �÷���ˮ����Ӳˮ����ˮ | |
| B�� | ��ʳ���ݿɳ�ȥ��ˮƿ�ڵ�ˮ�� | |
| C�� | �������õ�������ǰ�Ƚ��л��ʵ�� | |
| D�� | ��ȼ�պ�����ζ�ķ��������·������Dz�˿���Ǻϳ���ά���� |
18���������ʵ������뻯ѧʽ����ϵ��ǣ�������
| A�� | ���� NaOH �� | B�� | ��ʯ��Ca��OH��2 �� | ||
| C�� | С�մ� NaHCO3 �� | D�� | �ɱ� H2O ������ |