��Ŀ����
ij��ȤС����ʵ������ģ��������ѧԭ����ʵ�飬�Թ������ɷֵȽ���̽����
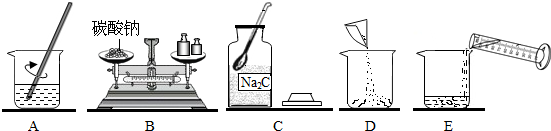
�����ʵ�顿��һ����̼��ԭ��������������ĩ��ʵ��װ����ͼ1��
������ʵ�顿��С�鰴����ʵ����ƣ���һ���¶��½�����ʵ�飬����������ݣ�
�ٷ�Ӧǰ�����ܺ���ʢ��������ĩ��������66.0g�������ܵ�����Ϊ60.0g��
�ڷ�Ӧ�����ܺ���ʢ��ɫ�����������65.8g����ȴ�����³�����
�����������ۡ���ʵ���У��������ڹ����ĩ�ɺ�ɫȫ����Ϊ��ɫ��С��ͬѧ��Ϊ�ú�ɫ����ȫ����������С��ͬѧ��������ɣ����Ǵ������������ϣ�
���������ϡ�
��CO��ԭFe2O3�Ĺ������ijɷ��뷴Ӧ�¶ȡ���Ӧʱ��������йأ�
��CO��ԭFe2O3��ʵ������й�����������Fe3O4��FeO��Fe��
��NaOH��Һ��ʯ��ˮ���ƣ�������CO2��������룮
��
��ʵ������롿
��1����С��ͬѧ����Ӧ��ĺ�ɫ������ĥ�����ô������������ֺ�ɫ�����ĩȫ���ܱ���������ú�ɫ�����ĩ�в����ܺ����������� ��
��2�����в��룺��ɫ�����ĩ���� a��ȫ��Ϊ����b�� ��c�� ��
���ó����ۡ�
��3��ͨ��ʵ���л�õ����ݽ��м��㣬�ó���ɫ�����ĩΪ��д���ƣ� ��
����˼�����ۡ�
��4��һ����̼��ԭ��������ʵ���У���Ҫ�õ������ʵ���߷�Ӧ�¶ȣ�
��5��ʵ��װ��ͼ�в������߿��ڵ�װ�ã��������� ��
A���ռ�CO B������CO2 C������CO2
��ʵ��Ӧ�á�
С��������ͬѧ��ⶨij������ʯ��Fe2O3��������������CO��10g������ʯ��Ʒ��ַ�Ӧ�����ʲ����뷴Ӧ�����������ɵ�������������NaOH��Һ��ȫ���գ�����Һ�������뷴Ӧʱ��ı仯��ϵ��ͼ2��
��6��������Ӧ����CO2������Ϊ g��
��7������ó�����ʯ�������������������� ������Ҫ�м�����̣�

�����ʵ�顿��һ����̼��ԭ��������������ĩ��ʵ��װ����ͼ1��
������ʵ�顿��С�鰴����ʵ����ƣ���һ���¶��½�����ʵ�飬����������ݣ�
�ٷ�Ӧǰ�����ܺ���ʢ��������ĩ��������66.0g�������ܵ�����Ϊ60.0g��
�ڷ�Ӧ�����ܺ���ʢ��ɫ�����������65.8g����ȴ�����³�����
�����������ۡ���ʵ���У��������ڹ����ĩ�ɺ�ɫȫ����Ϊ��ɫ��С��ͬѧ��Ϊ�ú�ɫ����ȫ����������С��ͬѧ��������ɣ����Ǵ������������ϣ�
���������ϡ�
��CO��ԭFe2O3�Ĺ������ijɷ��뷴Ӧ�¶ȡ���Ӧʱ��������йأ�
��CO��ԭFe2O3��ʵ������й�����������Fe3O4��FeO��Fe��
��NaOH��Һ��ʯ��ˮ���ƣ�������CO2��������룮
��
| �������� | ���������� | �������� | ������ | ���� |
| ��ɫ | ��ɫ | ��ɫ | ��ɫ | ��ɫ |
| �ܷ������� | �� | ���� | ���� | �� |
��1����С��ͬѧ����Ӧ��ĺ�ɫ������ĥ�����ô������������ֺ�ɫ�����ĩȫ���ܱ���������ú�ɫ�����ĩ�в����ܺ�����������
��2�����в��룺��ɫ�����ĩ���� a��ȫ��Ϊ����b��
���ó����ۡ�
��3��ͨ��ʵ���л�õ����ݽ��м��㣬�ó���ɫ�����ĩΪ��д���ƣ�
����˼�����ۡ�
��4��һ����̼��ԭ��������ʵ���У���Ҫ�õ������ʵ���߷�Ӧ�¶ȣ�
��5��ʵ��װ��ͼ�в������߿��ڵ�װ�ã���������
A���ռ�CO B������CO2 C������CO2
��ʵ��Ӧ�á�
С��������ͬѧ��ⶨij������ʯ��Fe2O3��������������CO��10g������ʯ��Ʒ��ַ�Ӧ�����ʲ����뷴Ӧ�����������ɵ�������������NaOH��Һ��ȫ���գ�����Һ�������뷴Ӧʱ��ı仯��ϵ��ͼ2��
��6��������Ӧ����CO2������Ϊ
��7������ó�����ʯ��������������������
���㣺ʵ��̽�����ʵ���ɳɷ��Լ�����,�������������Լ���Ӧ��,һ����̼��ԭ������,���ݻ�ѧ��Ӧ����ʽ�ļ���
ר�⣺��ѧ̽��
��������ʵ������롿��1�����������ɫ�����ĩȫ���ܱ��������������ܺ��е����ʣ�
��2�����������������������Ǻ�ɫ�����ܱ������������Ժ�ɫ��ĩ���в��룮
���ó����ۡ���3����������������������������������ͺ��е���Ԫ�ص��������ٸ��ݼ��ٵ�����������ɫ�����ĩ�ijɷ֣�
����˼�����ۡ���5������һ����̼�ж�����Ⱦ������Ҫ����β���Ͷ�����̼��������������Һ��Ӧ������
��6��NaOH��Һ���ӵ��������ڶ�����̼��������
��2�����������������������Ǻ�ɫ�����ܱ������������Ժ�ɫ��ĩ���в��룮
���ó����ۡ���3����������������������������������ͺ��е���Ԫ�ص��������ٸ��ݼ��ٵ�����������ɫ�����ĩ�ijɷ֣�
����˼�����ۡ���5������һ����̼�ж�����Ⱦ������Ҫ����β���Ͷ�����̼��������������Һ��Ӧ������
��6��NaOH��Һ���ӵ��������ڶ�����̼��������
����⣺��1���������֪����ɫ�����ĩȫ���ܱ���������ú�ɫ�����ĩ�в����ܺ���������������������
��2���������֪����ɫ�����ĩȫ���ܱ���������ɫ�����ĩ������ȫ��Ϊ����ȫ��Ϊ���������������������������۵Ļ���
���ó����ۡ���3����ʵ������ݿ�֪��������������Ϊ��66.0g-60.0g=6g����Щ�������к��е���Ԫ�ص�����Ϊ��6g��
=1.8g������������Ϊ��6g-1.8g=4.2g�����6g��������ȫ�������������ٵ�����Ӧ��1.8g���������֪��������ٵ�����Ϊ��66g-65.8g=0.2g�����ԣ�ʣ������������������Ϊ4.2g��1.6g=21��8����������������������
����˼�����ۡ���5������һ����̼�ж�����Ⱦ������Ҫ����β����������̼��������������Һ��Ӧ�����ԣ����߿��ڵ�װ�ã����������ռ�һ����̼�����ն�����̼��
��6����Ӧ����CO2������Ϊ106.6g-100.0g=6.6g
��7���⣺�����Ʒ��������������Ϊx
3CO+Fe2O3
2 Fe+3CO2?
160 132
X 6.6g
=
X=8.0g
������ʯ������������������
��100%=80%
����Ʒ������������������Ϊ80%��
�ʴ�Ϊ����1������������
��2��b��������������c������������������
��3��������������
��5��AB��
��6��6.6g��
��7��80%����2�֣�����Ҫ�н�����̣���
��2���������֪����ɫ�����ĩȫ���ܱ���������ɫ�����ĩ������ȫ��Ϊ����ȫ��Ϊ���������������������������۵Ļ���
���ó����ۡ���3����ʵ������ݿ�֪��������������Ϊ��66.0g-60.0g=6g����Щ�������к��е���Ԫ�ص�����Ϊ��6g��
| 16��3 |
| 56��2+16��3 |
����˼�����ۡ���5������һ����̼�ж�����Ⱦ������Ҫ����β����������̼��������������Һ��Ӧ�����ԣ����߿��ڵ�װ�ã����������ռ�һ����̼�����ն�����̼��
��6����Ӧ����CO2������Ϊ106.6g-100.0g=6.6g
��7���⣺�����Ʒ��������������Ϊx
3CO+Fe2O3
| ||
160 132
X 6.6g
| 160 |
| 132 |
| x |
| 6.6g |
X=8.0g
������ʯ������������������
| 8.0g |
| 10.0g |
����Ʒ������������������Ϊ80%��
�ʴ�Ϊ����1������������
��2��b��������������c������������������
��3��������������
��5��AB��
��6��6.6g��
��7��80%����2�֣�����Ҫ�н�����̣���
�����������ۺ��Խ�ǿ����ȫ��Ŀ�����һ����̼��ԭ���������������������ݶԻ������ж������������Ѷȣ����շ�Ӧǰ��Ԫ�ص��������䣬�����Ϸ���ʽ��������˳�����

��ϰ��ϵ�д�
 �̲�ȫ���ִʾ�ƪϵ�д�
�̲�ȫ���ִʾ�ƪϵ�д�
�����Ŀ
 С������������ʱ�������ȡ������������ͼ��ʾ����������Ȥ�����ԡ�̽����������������ͳɷ֡���Ϊ�о���ѧϰ�Ŀ��⣬С��ͨ�������й����ϻ�֪����������������Լռ
С������������ʱ�������ȡ������������ͼ��ʾ����������Ȥ�����ԡ�̽����������������ͳɷ֡���Ϊ�о���ѧϰ�Ŀ��⣬С��ͨ�������й����ϻ�֪����������������Լռ
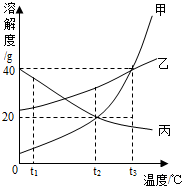 �ס��ҡ������ֹ������ʵ��ܽ��������ͼ��ʾ������ͼʾ�ش��������⣺
�ס��ҡ������ֹ������ʵ��ܽ��������ͼ��ʾ������ͼʾ�ش��������⣺