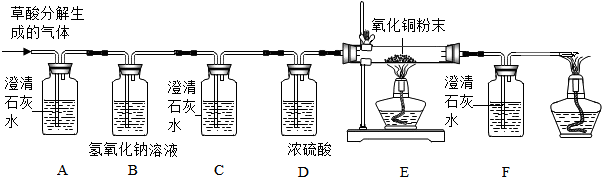
��Ŀ����
 ���ݻ�ͭ��ͭп�Ͻ��������Ͻ�Ԫ������IJ�ͬ����ͭ��Ϊ��ͨ��ͭ�������ͭ���֣�������ͭ��һ����ͨ��ͭ���������������ǣ�ʵ��С���ij�ֵ��ǵijɷֽ��з�����ȡ64.3g��Ʒ������300gϡ�����г���ܽ⣬������������ϡ���������仯��ϵ������ͼ��
���ݻ�ͭ��ͭп�Ͻ��������Ͻ�Ԫ������IJ�ͬ����ͭ��Ϊ��ͨ��ͭ�������ͭ���֣�������ͭ��һ����ͨ��ͭ���������������ǣ�ʵ��С���ij�ֵ��ǵijɷֽ��з�����ȡ64.3g��Ʒ������300gϡ�����г���ܽ⣬������������ϡ���������仯��ϵ������ͼ����1����ͼ֪�ܽ���Ʒ����ϡ�����������
��2��������Ʒ��ͭ��������������д��������̣��������lλС����
��3�����ܽ���Ʒ�����Һ�еμӼ��η�̪�������μ�10%��Na0H��Һ����Һǡ�ñ�Ϊ��ɫʱ���ĵ�NaOH��Һ������100g���ܽ���Ʒ��ϡ������Һ������������
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ���,�й��������������ļ���
ר�⣺�ۺϼ��㣨ͼ���͡������͡��龰�ͼ����⣩
��������1�����ݽ������˳��ɵ�֪����ͭ��ͭ����ϡ���ᷢ����Ӧ�����Ե���Ӧ�в��ٲ�������ʱ����Ӧ������
��2����Ʒ��ͭ����������Ʒ��֮�ȼ�Ϊ����ͭ��������������ͭ���μӷ�Ӧ��ͭ��������������ã���Ʒ����-����п������=��ͭ��Ʒ��ͭ��������������Ҫ���ݷų�
0.6g�����ȼ��������п��������
��3�����ݣ�2���з�Ӧ����ʽ�������0.6g������Ҫ��ϡ�������������������������ϡ����ķ�Ӧ����ʽ��������ϡ�����������
��2����Ʒ��ͭ����������Ʒ��֮�ȼ�Ϊ����ͭ��������������ͭ���μӷ�Ӧ��ͭ��������������ã���Ʒ����-����п������=��ͭ��Ʒ��ͭ��������������Ҫ���ݷų�
0.6g�����ȼ��������п��������
��3�����ݣ�2���з�Ӧ����ʽ�������0.6g������Ҫ��ϡ�������������������������ϡ����ķ�Ӧ����ʽ��������ϡ�����������
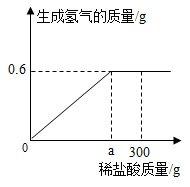
����⣺��1������ͼʾ֪����ϡ���������Ϊagʱ���������������ٱ仯�������ܽ���Ʒ����ϡ�����������ag��
���ag��
��2�������0.6gH2��Ҫп������Ϊx����Ҫ���������Ϊy����
Zn+2HCl�TZnCl2+H2��
65 71 2
x y 0.6g
=
x=19.5g��
=
y=21.3g��
��ͭ��Ʒ��ͭ����������=
��100%��69.7%��
����Ʒ��ͭ����������Լ��69.7%��
��3��100g 10%��Na0H��Һ����������Ϊ10g��
����10%��Na0H��Һ��Ӧ��Ҫ���������Ϊz����
NaOH+HCl�TNaCl+H2O
40 36.5
10g z
=
z=9.125g
��ϡ�������������Ϊ��
��100%��10%��
���10%��
���ag��
��2�������0.6gH2��Ҫп������Ϊx����Ҫ���������Ϊy����
Zn+2HCl�TZnCl2+H2��
65 71 2
x y 0.6g
| 65 |
| x |
| 2 |
| 0.6g |
| 71 |
| y |
| 2 |
| 0.6g |
��ͭ��Ʒ��ͭ����������=
| 64.3g-19.5g |
| 64.3g |
����Ʒ��ͭ����������Լ��69.7%��
��3��100g 10%��Na0H��Һ����������Ϊ10g��
����10%��Na0H��Һ��Ӧ��Ҫ���������Ϊz����
NaOH+HCl�TNaCl+H2O
40 36.5
10g z
| 40 |
| 10g |
| 36.5 |
| z |
��ϡ�������������Ϊ��
| 21.3g+9.125g |
| 300g |
���10%��
�����������ѶȲ�����Ҫ�����˸��ݻ�ѧ����ʽ�������йص������Ӷ�ʹѧ������ʶ��ѧ��Ӧ�ı��ʣ����뻯ѧ����ʽ��������ݱ����Ǵ�������������У�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
��ʡ��ũҵ��ʡ��ÿ���ո�ʱ��Щ������������սոѣ����¿�������һ�ȴﵽ�ض���Ⱦ�������йش����ո�˵��������ǣ�������
| A�����ýոѷ��Ͳ������� |
| B���ոѸ��ú��ת��Ϊ�л����� |
| C���ո�ȼ�ղ����Ķ�����̼������������Ҫԭ�� |
| D���ո�ȼ�ղ����Ĵ����̳������˿����еĿ���������� |
��ѧ�����彡�������й�ϵ�����жԡ���ѧ���������ʶ�в���ȷ���ǣ�������
| A������������ʳƷ��װ�����Է��� |
| B��ʹ�ü�����ǿ�����Ľ��������ڷ���ȱ����ƶѪ |
| C���������������Ͼ����ȹ��� |
| D���ü�ȩ����ˮ��Ʒ�ﵽ���ʵ�Ŀ�� |
 ijУ��ѧС��ͬѧ�Ӽ��д������ۡ�ʳ�Ρ�����ݴ�ۡ�ȥ�۷ۺͲ��ֱ��ʵ���ʯ��6�ֹ������ʣ���ʦ����ȡ������3�ֲ���ţ���С��ͬѧͨ��ʵ��ȷ�Ϸֱ�Ϊ���ֹ��壮
ijУ��ѧС��ͬѧ�Ӽ��д������ۡ�ʳ�Ρ�����ݴ�ۡ�ȥ�۷ۺͲ��ֱ��ʵ���ʯ��6�ֹ������ʣ���ʦ����ȡ������3�ֲ���ţ���С��ͬѧͨ��ʵ��ȷ�Ϸֱ�Ϊ���ֹ��壮