��Ŀ����
ij��ѧС��ͬѧ���۽ǶȽ�һ�����⸴�ֽⷴӦ��ʵ�ʡ�
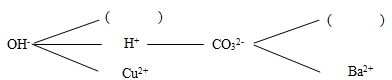
��1�����ֽⷴӦ��ʵ����ijЩ���������ӽ�����ɳ���������� �Ĺ��̡�
��2�����ݸ��ֽⷴӦ��ʵ�ʣ��γ�һ�����ֽⷴӦ����ͼ����ͼ��ֱ������������������֮���ܹ�������Ϸ�����Ӧ������ѡ����ʵ�����������ͼ�����ڣ�ʹ���������ʱ��һ�������������һ���г�����ע������������۵ijɷ���ͬ�����ɣ��������������ɵĻ�ѧ��Ӧ����ʽ������ ��
��1��ˮ ��2��NH4Cl+ NaOH �� Na Cl + H2O + NH3��(���������𰸾���) NH4 + �� Ca2+

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ