��Ŀ����
��֪��N a2CO3���Ȳ��ֽ⣬2NaHCO3
a2CO3���Ȳ��ֽ⣬2NaHCO3 Na2CO3��CO2����H2O��ij������Ʒ�л���������̼�����ƣ�Ϊ�˲ⶨ��Ʒ�Ĵ��ȣ���ѩͬѧ��������ʵ�飺ȷ��ȡ��Ʒ10.0 g�����Թ��У����ȳ�ַ�Ӧ��������ų�����
Na2CO3��CO2����H2O��ij������Ʒ�л���������̼�����ƣ�Ϊ�˲ⶨ��Ʒ�Ĵ��ȣ���ѩͬѧ��������ʵ�飺ȷ��ȡ��Ʒ10.0 g�����Թ��У����ȳ�ַ�Ӧ��������ų����� ����CO2����224 mL(��������������ܶ�Ϊ1.964 g/L)����ش��������⣺
����CO2����224 mL(��������������ܶ�Ϊ1.964 g/L)����ش��������⣺
(1)����CO2������Ϊ___g(��ȷ��0.01 g)��
(2)������Ʒ�е�Na2CO3�����������Ƕ��٣�(Ҫ��д��������̣������ȷ��0.1%)
(3)����a g�ô�����Ʒ��ּ�����������ų�����ȴ��Ƶ�ʣ�������������Ϊb g(b��a)����ô�����Ʒ��Na2CO3�����������ı���ʽ(Ҫ��д���������)��
0.44_
�⣺(2)�贿����Ʒ�е�NaHCO3������Ϊx
2NaHCO3 Na2CO3 ��CO2����H2O
Na2CO3 ��CO2����H2O
168������ ������������44
x 0.44 g
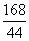 ��
��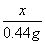
x��1.68 g
������Ʒ�е�Na2CO3����������Ϊ��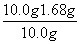 ��100%��83.2%
��100%��83.2%
(3)�贿����Na2CO3����������Ϊy
2NaHCO3 Na2CO3��CO2����H2O�����ٵ�����
Na2CO3��CO2����H2O�����ٵ�����
��168����������������44�� ��18�� 18��44��62
a g��(1��y)������������������������a g��b g
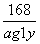 ��
��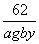
y��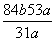 ��100%
��100%

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д� ��ʹ��ɫʯ����Һ����
��ʹ��ɫʯ����Һ���� �ֵ��ǣ� ����
�ֵ��ǣ� ���� P+
P+