��Ŀ����
7����NaOH��Ca��0H��2��Һ��ѡ�����ʣ��û�ѧ����ʽ��ʾ����1������C0�к�CO2��Ca��0H��2��
��2����ȥCO�е�CO2��NaOH��
�ش���������⣺
�������ƺ��������ƶ��ǰ�ɫ���壬���ܷ��ü����������������ֿ�����
����������Һ��ʯ��ˮ�������ն�����̼������ȥ������̼��������������Һ�������������̼һ��Ҫ�ó���ʯ��ˮ��ԭ�����������Ƶ��ܽ�ȱ��������ƴ�����̼��ʹ����ʯ��ˮ����ǣ�
���� ������̼��ʹ����ʯ��ˮ����ǣ�
�������ƺͶ�����̼��Ӧ����̼���ƺ�ˮ��
һ�������£��������Ƶ��ܽ��ԶԶ�����������Ƶ��ܽ�ȣ�
���������ܹ����տ����е�ˮ���������������Ʋ��ܣ�
��� �⣺��1����Ϊ������̼��ʹ����ʯ��ˮ����ǣ���˳��ó���ʯ��ˮ����C0�к�������̼��
���Ca��0H��2��
��2����Ϊ���������ܹ����ն�����̼���壬��˳�������������Һ��ȥCO�еĶ�����̼��
���NaOH��
�������ƺ��������ƶ��ǰ�ɫ���壬�������ߵ��������������ǣ����������������ƹ��塢�������ƹ���ֱ���������������У�һ��ʱ���۲죬�ܹ���������������ƣ����ܳ�������������ƣ�
��Ϊ�������Ƶ��ܽ�ȱ��������ƴ�����̼��ʹ����ʯ��ˮ����ǣ����Գ�ȥ������̼��������������Һ�������������̼һ��Ҫ�ó���ʯ��ˮ��
����������Ƶ��ܽ�ȱ��������ƴ�����̼��ʹ����ʯ��ˮ����ǣ�
���� �������ʵ�飬��ѧ�ؽ���ʵ�顢����ʵ�飬�ǵó���ȷʵ����۵�ǰ�ᣬ���Ҫѧ�����ʵ�顢����ʵ�顢����ʵ�飬Ϊѧ�û�ѧ֪ʶ�춨������

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
17����158g���������ȡ������ʵ�����Ӧһ��ʱ�����ǰ�������������Ԫ�ص���������֮��Ϊ75��79�������������������ǣ�������
| A�� | 8g | B�� | 16g | C�� | 32g | D�� | 4g |
18�����й���2H2+O2 $\frac{\underline{\;��ȼ\;}}{\;}$ 2H2O��˵������ȷ���ǣ�������
| A�� | ��ʾ��������������ˮ | |
| B�� | ��ʾ�����������ڵ�ȼ����������ˮ | |
| C�� | ��ʾÿ4��������������32��������������ȫ��Ӧ����36��������ˮ | |
| D�� | ��ʾ������������ˮ������Ŀ��Ϊ2��1��2 |
2�����з�Ӧ�У���������ԭ��Ӧ���������û���Ӧ���ǣ�������
| A�� | Fe+2HCl��FeCl2+H2�� | B�� | H2+Cu��Cu+H2O | ||
| C�� | Fe2O3+3CO��2Fe+3CO2 | D�� | Fe+CuSO4��Cu+FeSO4 |

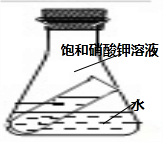 ����ͬѧ�������ͼ��ʾ��װ�ý���Ȥζʵ�飮
����ͬѧ�������ͼ��ʾ��װ�ý���Ȥζʵ�飮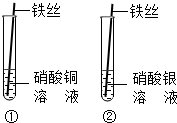 ij����С���ͬѧ�ԡ�����ͭ�����Ļ�ѧ���ʡ�Ϊ���⣬���п�ѧ̽������������ѧ֪ʶ�������̽�����⣮
ij����С���ͬѧ�ԡ�����ͭ�����Ļ�ѧ���ʡ�Ϊ���⣬���п�ѧ̽������������ѧ֪ʶ�������̽�����⣮