��Ŀ����
20��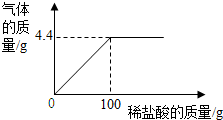 Ϊ����Na2CO3��NaCl��ɵ���Ʒ��NaCl������������ȡ15g�ù�����Ʒ��ȫ������200mLˮ�У������õ��Ļ����Һ����ε���������������Ϊ7.3%��ϡ���ᣬ��¼����ͼ��ʾ�����߹�ϵ����ˮ���ܶ�Ϊ1g/mL������������������ȷ��0.1%��
Ϊ����Na2CO3��NaCl��ɵ���Ʒ��NaCl������������ȡ15g�ù�����Ʒ��ȫ������200mLˮ�У������õ��Ļ����Һ����ε���������������Ϊ7.3%��ϡ���ᣬ��¼����ͼ��ʾ�����߹�ϵ����ˮ���ܶ�Ϊ1g/mL������������������ȷ��0.1%����1����Na2CO3��ϡ����ǡ����ȫ��Ӧʱ������7.3%��ϡ����������Ƕ��٣�
��2����Ʒ��NaCl�����������Ƕ��٣�
��3������Na2CO3��ϡ����ǡ����ȫ��Ӧʱ��������Һ����������������
���� ��1������ͼʾ���ҳ��ں�ʱ��Ӧ����ȫ������֪����������̼����ǡ����ȫ��Ӧʱ�����������������
��2����Ϊ̼���������ᷴӦ���ɶ�����̼���壮���ݷ�Ӧ�Ļ�ѧ����ʽ������������������г�����ʽ���Ϳɼ������Ʒ��̼���Ƶ�������Ȼ����ݡ���Ʒ����-̼���Ƶ�����=��������Ȼ��Ƶ����������㼴�ɣ�
��3����������̼����ǡ����ȫ��Ӧʱ��������ҺΪ�Ȼ�����Һ�����������Ȼ�������Ϊ��Ʒ���Ȼ����뷴Ӧ�����Ȼ��Ƶ������ͣ����������غ㶨�ɣ���÷�Ӧ����Һ��������ʹ����Һ�����ʵ������������㹫ʽ����������Һ�����ʵ�����������
��� �⣺��1����ͼʾ��֪���������ᵽ100gʱ�����岻�����ɣ�˵����Ӧ����ȫ��
�ʴ�Ϊ��100g��
��2������Ʒ��̼���Ƶ�����Ϊx�������Ȼ��Ƶ�����Ϊy��
Na2CO3+2HCl�T2NaCl+CO2��+H2O
106 117 44
x y 4.4g
$\frac{106}{x}=\frac{117}{y}=\frac{44}{4.4g}$
x=10.6 g y=11.7g
��Ʒ��NaCl����������=$\frac{15g-10.6}{15g}��$100%=29.3%
��3��Na2CO3��ϡ����ǡ����ȫ��Ӧʱ��������ҺΪ�Ȼ�����Һ����������Ϊ11.7g+15g-10.6g=16.1g����Һ������=15g+200g+100g-4.4g=310.6g
��Һ��NaCl%=$\frac{16.1g}{310.6g}$��100%=5.2%��
�𰸣�
��1��100g��
��2����Ʒ��NaCl������������29.3%��
��3��������Һ��������������Ϊ5.2%��
���� ������Ҫ����ѧ������ȫ��Ӧ����ʶ���Լ����û�ѧ����ʽ����������������ʽ���м����������������ѧ�����������˼ά��ȣ�ǿ����ѧ������֪ʶ��������

| A�� | �ɱ������˹����� | |
| B�� | CO2������������ˮ��̼������ | |
| C�� | CO2�μ�ֲ��Ĺ�����ÿ����ũ������� | |
| D�� | CO2������� |
| A�� | ZnCl2 | B�� | AlCl3 | C�� | MgCl2 | D�� | FeCl3 |
| A�� | ���� | B�� | ���� | C�� | ������̼ | D�� | ϡ������ |
| A�� | ��ʳ�׳�ˮ�����ϵ�ˮ�� | B�� | �û���̿����Ȼˮ�еij�ζ���� | ||
| C�� | �������ȼ����̿�� | D�� | �������ᡢС�մ�ˮ��������ˮ |
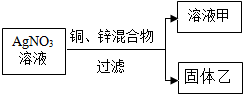
 ����A��B��C��D��E��F��G�������ʣ�C��F������Ľ�����B�����嵥�ʣ�DΪdz��ɫ��Һ��GΪ��ɫ��Һ������֮��������¹�ϵ��
����A��B��C��D��E��F��G�������ʣ�C��F������Ľ�����B�����嵥�ʣ�DΪdz��ɫ��Һ��GΪ��ɫ��Һ������֮��������¹�ϵ�� Ϊ�˲ⶨ�����������ĺ�����С����������·�����ѡ��ʵ���ܼ�Ϊ40ml��ֱ����������Ӧ�������������İ������Թܣ�����Ƥ����ֱ�����ܣ�ͨ��������ʵ���ݻ�Ϊ60mL�������Ժܺõ���Ͳע���������ͼ��ʾ��ʵ��װ�ã������װ���ܰ���С���������������У��Ұ�����ռ����뵼���ڵ�����������Բ��ƣ���ش��������⣺
Ϊ�˲ⶨ�����������ĺ�����С����������·�����ѡ��ʵ���ܼ�Ϊ40ml��ֱ����������Ӧ�������������İ������Թܣ�����Ƥ����ֱ�����ܣ�ͨ��������ʵ���ݻ�Ϊ60mL�������Ժܺõ���Ͳע���������ͼ��ʾ��ʵ��װ�ã������װ���ܰ���С���������������У��Ұ�����ռ����뵼���ڵ�����������Բ��ƣ���ش��������⣺