��Ŀ����
ijѧУ��ѧ�о���ѧϰС���ͬѧ���ⶨij������Ʒ�����Ȼ��ƣ���̼���Ƶĺ���������ȡ16.6g����Ʒ����μ���ϡ����ֱ�����ٲ�������Ϊֹ������ȥϡ����90g��ʵ���в���t1��t5�������������������������ܶȰ�1.97g/L���㣩���±���ʾ��
��1��������ݲⶨ�����ݷ�����______���Ӻ���Ʒ��̼�����Ѿ���Ӧ��ȫ��
��2���������Ʒ��̼���Ƶ�����������
��3�����㷴Ӧ��������Һ�����ʵ�����������
| ʱ��/���� | t1 | t2 | t3 | t4 | t5 |
| ���/L | 1.52 | 2.54 | 3.05 | 3.35 | 3.35 |
��2���������Ʒ��̼���Ƶ�����������
��3�����㷴Ӧ��������Һ�����ʵ�����������
��1���ɱ��е����ݿ�֪��t4���Ӻ���������������������ʱ��˵������Ʒ��̼�����Ѿ���Ӧ��ȫ��
��2�����ɶ�����̼������Ϊ��3.35L��1.97g/L=6.6g��
������6.6g������̼��ҪNa2CO3������Ϊx��ͬ�����ɵ��Ȼ��Ƶ�����Ϊy
Na2CO3+2HCl�T2NaCl+CO2��+H2O
106 117 44
x y 6.6g
=
=
��ã�x=15.9g y=17.55g
��Ʒ��̼���Ƶ���������Ϊ��
��100%=95.8%
��3����Ӧ��������Һ�����ʵ���������Ϊ��
��100%=18.3%
�ʴ�Ϊ����1��t4����2����Ʒ��Na2CO3����������Ϊ95.8%����3����Ӧ����Һ�����ʵ���������ԼΪ18.3%��
��2�����ɶ�����̼������Ϊ��3.35L��1.97g/L=6.6g��
������6.6g������̼��ҪNa2CO3������Ϊx��ͬ�����ɵ��Ȼ��Ƶ�����Ϊy
Na2CO3+2HCl�T2NaCl+CO2��+H2O
106 117 44
x y 6.6g
| 106 |
| x |
| 117 |
| y |
| 44 |
| 6.6g |
��ã�x=15.9g y=17.55g
��Ʒ��̼���Ƶ���������Ϊ��
| 15.9g |
| 16.6g |
��3����Ӧ��������Һ�����ʵ���������Ϊ��
| 17.55g+(16.6g-15.9g) |
| 16.6g+90g-6.6g |
�ʴ�Ϊ����1��t4����2����Ʒ��Na2CO3����������Ϊ95.8%����3����Ӧ����Һ�����ʵ���������ԼΪ18.3%��

��ϰ��ϵ�д�
�����Ŀ
����ʹ�õ���ˮ���ײ���һ��ˮ������Ҫ�ɷ���̼��ƺ�������þ����ϡ����������ˮ����ijѧУ��ѧ�о���ѧϰС���ͬѧͨ��ʵ��ⶨˮ����̼��Ƶĺ�������������ϡ������뵽400gˮ���У��Ѳ�����CO2������������NaOH��Һ���գ�ͬʱ����3������NaOH��Һ���ӵ���������������ʾ��
��1����������ֽ�ϣ��Է�Ӧʱ��Ϊ�����꣬��NaOH��Һ���ӵ�����Ϊ�����꣬�����ܹ�������������������淴Ӧʱ��仯���ɵĹ�ϵ���ߣ���д��CO2���屻����NaOH��Һ���յĻ�ѧ����ʽ
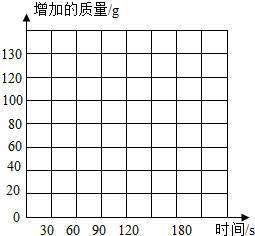
��2���ӱ��п�����NaOH��Һ��������132g�������������ӣ����� ������ˮ����̼��Ƶ�������������д���ļ�����̣�
| ʱ��/s | 0 | 30 | 60 | 90 | 120 | 150 | 180 |
| ���ӵ�����/g | 0 | 60 | 100 | 120 | 132 | 132 | 132 |

��2���ӱ��п�����NaOH��Һ��������132g�������������ӣ�����
ijѧУ��ѧ�о���ѧϰС���ͬѧ���ⶨij������Ʒ�����Ȼ��ƣ���̼���Ƶĺ���������ȡ16.6g����Ʒ����μ���ϡ����ֱ�����ٲ�������Ϊֹ������ȥϡ����90g��ʵ���в���t1��t5�������������������������ܶȰ�1.97g/L���㣩���±���ʾ��
| ʱ��/���� | t1 | t2 | t3 | t4 | t5 |
| ���/L | 1.52 | 2.54 | 3.05 | 3.35 | 3.35 |
��2���������Ʒ��̼���Ƶ�����������
��3�����㷴Ӧ��������Һ�����ʵ�����������
����ʹ�õ���ˮ���ײ���һ��ˮ������Ҫ�ɷ���̼��ƺ�������þ����ϡ����������ˮ����ijѧУ��ѧ�о���ѧϰС���ͬѧͨ��ʵ��ⶨˮ����̼��Ƶĺ�������������ϡ������뵽400gˮ���У��Ѳ�����CO2������������NaOH��Һ���գ�ͬʱ����3������NaOH��Һ���ӵ���������������ʾ��
��1����������ֽ�ϣ��Է�Ӧʱ��Ϊ�����꣬��NaOH��Һ���ӵ�����Ϊ�����꣬�����ܹ�������������������淴Ӧʱ��仯���ɵĹ�ϵ���ߣ���д��CO2���屻����NaOH��Һ���յĻ�ѧ����ʽ______
��2���ӱ��п�����NaOH��Һ��������132g�������������ӣ�����______������ˮ����̼��Ƶ�������������д���ļ�����̣�
| ʱ��/s | 30 | 60 | 90 | 120 | 150 | 180 | |
| ���ӵ�����/g | 60 | 100 | 120 | 132 | 132 | 132 |

��2���ӱ��п�����NaOH��Һ��������132g�������������ӣ�����______������ˮ����̼��Ƶ�������������д���ļ�����̣�