��Ŀ����
��ҵ�����г�����ʳ�Σ���Ҫ�ɷ�ΪNa2CO3������ΪNaCl����ij������Ϊ�˲ⶨһ��������Na2CO3���������������������·���ʵ�飺ȡ������Ʒ�����ձ��У���ˮ����ܽ⣬�����μ�ϡ���ᣬ������������Ϊֹ���й����������ʾ����
��1����Ӧ����ʱ������������̼��������
��2��������Ʒ��Na2CO3����������������������ȷ��0.1%����ͬ��
��3����Ӧ���ձ�����Һ����������������
| ���� | ��Ʒ���� | ˮ������ | ����ϡ�������� | ��Ӧ����Һ���� |
| ������g�� | 22 | 50 | 56.8 | 120 |
��2��������Ʒ��Na2CO3����������������������ȷ��0.1%����ͬ��
��3����Ӧ���ձ�����Һ����������������
���㣺���ݻ�ѧ��Ӧ����ʽ�ļ���,�й��������������ļ���
ר�⣺�ۺϼ��㣨ͼ���͡������͡��龰�ͼ����⣩
��������1������̼���ƺ�ϡ���ᷴӦ�����Ȼ��ơ�ˮ��������̼�����������غ㶨�ɿ�֪��Ӧǰ����������ľ������ɵĶ�����̼��
��2��Ȼ����ݻ�ѧ����ʽ�����̼���Ƶ��������������̼���Ƶ�����������
��3�����÷�Ӧ����Һ���Ȼ��Ƶ���Һ������Na2CO3+2HCl�T2NaCl+CO2��+H2O����Ȼ��Ƶ�����������������������=
��100%������
��2��Ȼ����ݻ�ѧ����ʽ�����̼���Ƶ��������������̼���Ƶ�����������
��3�����÷�Ӧ����Һ���Ȼ��Ƶ���Һ������Na2CO3+2HCl�T2NaCl+CO2��+H2O����Ȼ��Ƶ�����������������������=
| �������� |
| ��Һ���� |
����⣺��1�����������غ㶨�ɵ�CO2=22+50+56.8-120=8.8g
��2�������ɵ�Na2CO3������ΪX�����ɵ�NaCl������ΪY����
Na2CO3+2HCl�T2NaCl+CO2��+H2O
106 117 44
X Y 8.8g
=
��
=
x=21.2g��y=23.4g
Na2CO3����������=
��100%=96.4%��
��3��NaCl����������=
��100%=20.2%
�𣺣�1������������̼������8.8g��
��2��Na2CO3������������96.4%��
��3����Ӧ���ձ�����Һ��������������20.2%��
��2�������ɵ�Na2CO3������ΪX�����ɵ�NaCl������ΪY����
Na2CO3+2HCl�T2NaCl+CO2��+H2O
106 117 44
X Y 8.8g
| 106 |
| x |
| 44 |
| 8.8g |
| 117 |
| y |
| 44 |
| 8.8g |
x=21.2g��y=23.4g
Na2CO3����������=
| 21.2g |
| 22g |
��3��NaCl����������=
| 23.4g+(22g-21.2g) |
| 120g |
�𣺣�1������������̼������8.8g��
��2��Na2CO3������������96.4%��
��3����Ӧ���ձ�����Һ��������������20.2%��
������ֻҪ���巴Ӧǰ��������������Ϊ�����˶�����̼����ļ��ɸ��ݷ�Ӧ�Ļ�ѧ����ʽ�����⣬��Һ�ͻ�ѧ��Ӧ�ںϵ���Ŀ�����ۺ��Ե����ͣ�Ҫ��Ƚϸߣ���

��ϰ��ϵ�д�
�����Ŀ
 NO��NO2�������в�ͬ���ʣ�ͨ������£�NO����ɫ���壬������ˮ��NO2�Ǻ���ɫ���壬������ˮ����ͼ����һ֧����NO2������Թܵ�����ʢˮ���ձ��У�NO2��ˮ��Ӧ���������һ��������������Ԥ�ⷴӦ��ȫ����ܳ��ֵ�ʵ������
NO��NO2�������в�ͬ���ʣ�ͨ������£�NO����ɫ���壬������ˮ��NO2�Ǻ���ɫ���壬������ˮ����ͼ����һ֧����NO2������Թܵ�����ʢˮ���ձ��У�NO2��ˮ��Ӧ���������һ��������������Ԥ�ⷴӦ��ȫ����ܳ��ֵ�ʵ������

 ��ҩ������Τע��Һ����������H7N9�����У���˵�����в���������ͼ��
��ҩ������Τע��Һ����������H7N9�����У���˵�����в���������ͼ��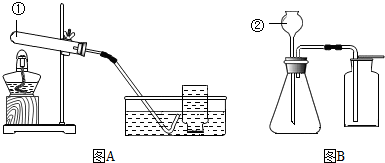 ������ͼ��ʾװ�ûش��������⣮
������ͼ��ʾװ�ûش��������⣮