��Ŀ����
9����ͼ��ʾ��ʵ������ȡ���峣�õ�ʵ��װ�ã��ش��������⣺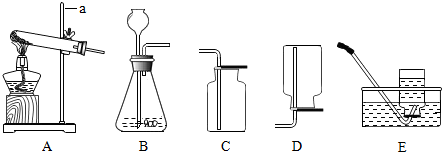
��1������a������������̨��
��2��ʵ�����ô���ʯ��ϡ���ᷴӦ��ȡCO2�Ļ�ѧ����ʽ��CaCO3+2HCl�TCaCl2+H2O+CO2������Ҫ��ȡ22��CO2��Ҫ50��CaCO3�μӷ�Ӧ��
��3��ѡA��Eװ����KMnO4������ʱ��E��ˮ����ԭ���Թܿ�û�����ţ�������ط�ĩ������������˳�ŵ��ܽ���ˮ�ۣ�ʹˮ���ڵ�ˮ����Ϻ�ɫ��
��4��ѡ�����巢��װ��ʱ��Ӧ���ǵ������Ƿ�Ӧ���״̬�ͷ�Ӧ������
���� ��1��Ҫ��Ϥ�������������ơ���;��ʹ�÷�����
��2��ʵ����ͨ���ô���ʯ��ʯ��ʯ��ϡ���ᷴӦ��ȡ������̼����Ӧ����Ҫ���ȣ�����ʯ��ʯ��ʯ����Ҫ�ɷ���̼��ƣ��ܺ�ϡ���ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼��
��3��ʵ�����ø��������ȡ����ʱ��Ҫ���Թܿڷ�һ����ȥ������
��4�����ݷ�Ӧ���״̬�ͷ�Ӧ����ѡ����װ�ã���������ͺ�Һ�����ͣ�
��� �⣺��1��a������̨��
��2��̼��ƺ�ϡ���ᷴӦ�Ļ�ѧ����ʽΪ��CaCO3+2HCl�TCaCl2+H2O+CO2����
����Ҫ̼��Ƶ�����Ϊx��
CaCO3+2HCl�TCaCl2+H2O+CO2����
100 44
x 22g
$\frac{100}{x}=\frac{44}{22g}$
x=50g
��3��ʵ�����ø��������ȡ����ʱ���Թܿ�û�����ţ�������ط�ĩ������������˳�ŵ��ܽ���ˮ�ۣ�ʹˮ���ڵ�ˮ����Ϻ�ɫ��
��4����ѡ�����巢��װ��ʱӦ���ǵ������ǣ���Ӧ���״̬�ͷ�Ӧ������
�𰸣�
��1������̨��
��2��CaCO3+2HCl�TCaCl2+H2O+CO2����50��
��3���Թܿ�û�����ţ�������ط�ĩ������������˳�ŵ��ܽ���ˮ�ۣ�ʹˮ���ڵ�ˮ����Ϻ�ɫ��
��4����Ӧ���״̬�ͷ�Ӧ������
���� ������Ҫ������������;����ѧ����ʽ����д��ʵ��װ�õ�ѡ��ѡ����װ��ʱ��Ҫ���Ƿ�Ӧ���״̬����Ӧ���������أ�ѡ���ռ�װ��ʱ��Ҫ���������ˮ���ԡ��ܷ��ˮ������ѧ��Ӧ���ܶȡ��ܷ�Ϳ����е����ʷ�����ѧ��Ӧ�����أ�

| A�� | ��������ּ����14��ԭ�� | |
| B�� | ����������̼Ԫ�ص���������Ϊ41.4% | |
| C�� | ����������̼����Ԫ�ص�������Ϊ6��1 | |
| D�� | ����Ũ����ζ����������ˮ�������������Ļ�ѧ���� |
| A�� | 3H--3����Ԫ�� | B�� | O2--������ | ||
| C�� | Al2O3--������ | D�� | 2N2--2���������� |
 Ԫ�����ڱ���þԪ�ؼ���ԭ�ӽṹʾ��ͼ��ͼ��ʾ���ɴ��ж�����˵��������ǣ�������
Ԫ�����ڱ���þԪ�ؼ���ԭ�ӽṹʾ��ͼ��ͼ��ʾ���ɴ��ж�����˵��������ǣ�������| A�� | þԪ�ص����ԭ������Ϊ24 | B�� | þ���ڽ���Ԫ�� | ||
| C�� | þԭ�ӵ�ԭ�Ӻ�����12������ | D�� | þԭ�����õ����� |
��1��Ϊ��Ѱ�������������۵ķ�������ѧ��ȤС���ͬѧ��������ͼ1ʵ�飺

ʵ��һ���ֱ�ȡ������������������֧�Թ��У���һ������ϡ���Ტ������������ͨ�����ʯ��ˮ�У����ֳ���ʯ��ˮ������ǣ�˵���������۾�����̼��ƣ�CaCO3����
ʵ������ֱ�ȡ������������������Ƭ�����գ���������������ŵ��ս���ë��ζ���������δ�ŵ��ս���ë��ζ���ɴ˿ɵó����ۣ���������в��������ʣ�
��2��Ϊ�˱Ƚ�����������̼��Ƶĺ�����������������ʵ�飬ÿ�θ�ȡ5g�������ۣ��ֱ�������ͼ2ʵ��װ���У�ϡ��������������������ֻ��̼�����ϡ����ᷴӦ�������壩���ⶨ������������������±���
| ��Ʒ�������mL���� | ��һ�� | �ڶ��� | ������ |
| ������� | 117.50 | 117.28 | 117.05 |
| ������� | 111.52 | 111.66 | 111.86 |
��ʵ�����ݿ�֪�����������̼��Ƶĺ�����������ۣ������������������=������
�ڸ������β��������ȡƽ��ֵ�����5g����������ɵ���������Ϊ0.22g�������������Ʒ��CaCO3������������
| A�� | C2H6 | B�� | C7H14 | C�� | C8H17OH | D�� | C6H12O6 |
 Ԫ�����ڱ���ѧϰ���о���ѧ����Ҫ���ߣ��ش��������⣺
Ԫ�����ڱ���ѧϰ���о���ѧ����Ҫ���ߣ��ش��������⣺ ����ԭ���ڻ�ѧ��Ӧ���õ�����õ�����ʧȥ�������ӣ�
����ԭ���ڻ�ѧ��Ӧ���õ�����õ�����ʧȥ�������ӣ�