��Ŀ����
����5�£��������ط����ݡ�������Ϊ����ĩ���������ͭ����ʱ��2500�꣬�������һ����ɫ���⣬���Ժ�����ˣ��з漫�����������������࣬��ë�Ϸ���
��ϸ�Ķ��������֣�����������⣺
��1����ͭ�ǽ��������� ������Ҫ�ɷ��� ������ţ���
��2����������Ϣ���Կ�������ͭ���� ��
��3���������������ɫ���⣬����Ҫ�ɷ�Ϊ��ʽ̼��ͭ[��ѧʽ��Cu2(OH)2CO3]�������ɫ�����γɵĿ���ԭ���� ��
�Ͻ� Cu��Zn
A��C
ͭ������е�������ˮ�֡�������̼�����ʷ�����Ӧ��Ӧ

 ���Ͱ�ͨ��ĩ���ϵ�д�
���Ͱ�ͨ��ĩ���ϵ�д�2011��9��29��21ʱ���칬һ�š�Ŀ���������
��Ȫ���Ƿ������ķ��䣨����ͼ��ʾ���������й����ռ�ʵ���ң����ֱ������۰˺š����۾źš�����ʮ�ŷɴ��Խӣ��Ӷ�������һ���й��ռ�ʵ���ҡ���ṹ��Ҫ�����ⲿ���塢ʵ��ա���Դ�ա�ȼ��ȼ��ϵͳ���Խӻ�����̫���ܵ��ȣ������������������µ�ʯī�ߡ�����֡��ѡ�����ͭ�ȵȡ��Իش��������⣺
�Ų�������� ��������������������
�Ƶ���ͭ�Ľ������ �������Ľ�����ԣ�����ڡ����� С�ڡ��������ڡ�����������Ƽ�ʵ����֤��ֻҪд���������û�ѧ����ʽ��ʾ�� ��
|
�ѵ�ԭ����TiCl4+4Na ==== Ti+4NaCl���÷�Ӧ�� �� ���������Ӧ���ͣ�����Ӧ��TiCl4����Ԫ�صĻ��ϼ�Ϊ ���Ȼ������� ������ӡ�����ԭ�ӡ������ӡ������ɵġ�
�� ���������Ӧ���ͣ�����Ӧ��TiCl4����Ԫ�صĻ��ϼ�Ϊ ���Ȼ������� ������ӡ�����ԭ�ӡ������ӡ������ɵġ�
����ͨ��ѧϰ֪�������и��ɷ��������������78%������21%��������̼ռ0.03%��
ϡ������ռ0.94%������0.03%����ô����������������������ھ�����һϵ�е�ѭ�����������������庬����Щ�ɷ��ء�
�������ϣ��˺�����������ɷ��������������78%������16%��������̼ռ4%��
ϡ������ռ1%��ˮ�����������ɷ�ռ1%��
�������ʵ��֤�����������к���������������̼��ˮ������
���� | ���� | ���� |
|
| ��������������� |
|
| �� |
|
| ���������к���ˮ���� |
 �������к��ж�����̼
�������к��ж�����̼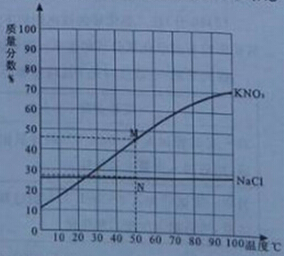
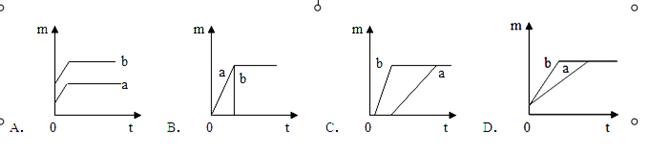
 ��ȡ�������Բ��ü�������أ���ɫ���壩�Ͷ������̻����ķ������ּ���������ȵ���������أ�һ���л��������Ķ������̣�b���ߣ����ֱ�ͬʱ���ȣ��ų�������������m���뷴Ӧʱ�䣨t���Ĺ�ϵͼ����ȷ���ǣ�������
��ȡ�������Բ��ü�������أ���ɫ���壩�Ͷ������̻����ķ������ּ���������ȵ���������أ�һ���л��������Ķ������̣�b���ߣ����ֱ�ͬʱ���ȣ��ų�������������m���뷴Ӧʱ�䣨t���Ĺ�ϵͼ����ȷ���ǣ�������