��Ŀ����
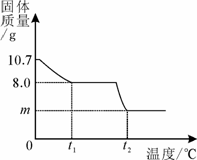 �������岿��ʧˮ��õ�ij����ͭ���壨CuSO4��xH2O������ѩͬѧΪ�о�CuSO4��xH2O���Ⱥ��������ʵ���ɣ���������ʵ�飺ȡ10.7gCuSO4��xH2O�������ʹ��ֽ⣬���Ƴ�������������¶ȵı仯��ϵͼ����ͼ��ʾ����t1��ʱ�ù�����ȫʧȥ�ᾧˮ����ѧ����ʽΪ��CuSO4��xH2O
�������岿��ʧˮ��õ�ij����ͭ���壨CuSO4��xH2O������ѩͬѧΪ�о�CuSO4��xH2O���Ⱥ��������ʵ���ɣ���������ʵ�飺ȡ10.7gCuSO4��xH2O�������ʹ��ֽ⣬���Ƴ�������������¶ȵı仯��ϵͼ����ͼ��ʾ����t1��ʱ�ù�����ȫʧȥ�ᾧˮ����ѧ����ʽΪ��CuSO4��xH2O  CuSO4+ xH2O��
CuSO4+ xH2O��
��1�����������ͭ���壨CuSO4��xH2O����x��ֵ��
��CuSO4��x H2O����Է�������Ϊ��160+18x��
��2��CuSO4��ͭԪ�ص����������Ƕ��٣�
��3�����µ�t2�棬���������Ǻ�ɫ���ʣ���m= g��
��д����m�Ĺ��̣�
��1�� CuSO4��xH2O  CuSO4+ xH2O��
CuSO4+ xH2O��
160 18x
8.0g ��10.7-8.0��g
160��8.0 g =18x��(10.7-8.0)g ��ã�x=3 ��2�֣�
��2��CuSO4��ͭԪ�ص���������=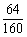 ��100%=40% ��2�֣�
��100%=40% ��2�֣�
��3��������֪�����µ�t2��ʱ�������ĺ�ɫ����ΪCuO�����������غ㶨�ɣ�CuSO4��CuO֮��������¹�ϵ��CuSO4��CuO[��Դ:ѧ,��,��Z,X,X,K]
160 80
8.0g m g
160��8.0 g=80��m g ��ã�m=4.0 ��2�֣�

 �����ҵ���������ϵ�д�
�����ҵ���������ϵ�д� �����̸�Ӯ����ٸ�Ч�����ܸ�ϰ���ϿƼ�������ϵ�д�
�����̸�Ӯ����ٸ�Ч�����ܸ�ϰ���ϿƼ�������ϵ�д� �����ҵ�����������ѧ���ӳ�����ϵ�д�
�����ҵ�����������ѧ���ӳ�����ϵ�д�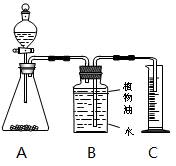 ���飺ȡ��
���飺ȡ�� ��̼��Ʒ�ĩ5g����ƿ�У���20g��������Һ�����Һ©�������òⶨ����������̼�������20�桢������ѹ����������Ũ�ȡ�װ
��̼��Ʒ�ĩ5g����ƿ�У���20g��������Һ�����Һ©�������òⶨ����������̼�������20�桢������ѹ����������Ũ�ȡ�װ С��ͬѧ��̽��ʵ������̼��ƺ�������ȡ������̼���Һ�����ʵijɷ֣�����һͬ����̽�����ش�����ϵ��������߸����⡣
С��ͬѧ��̽��ʵ������̼��ƺ�������ȡ������̼���Һ�����ʵijɷ֣�����һͬ����̽�����ش�����ϵ��������߸����⡣ �� �� ����ܡ����ܡ���֤�����IJ��룬�������� ��
�� �� ����ܡ����ܡ���֤�����IJ��룬�������� ��