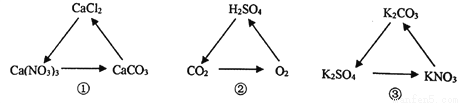
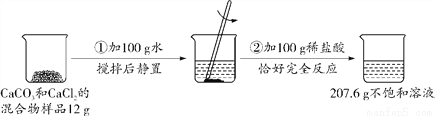
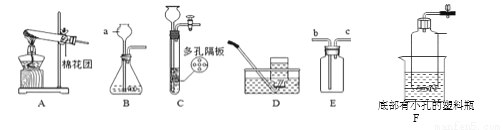
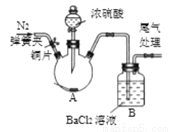
��Ŀ����
ij��ȤС����̽��Zn��Cu��Ag��R���ֽ����Ļ��˳��(RΪδ֪����)������������ʵ�飺
 ��������
��������
ͼA������������������������������ͼB
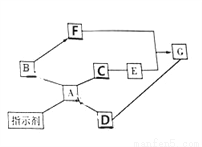
(1)��ͼA��ʾ�����ĸ�����˿ͬʱ�����ձ��У������з�����Ӧ�Ļ�ѧ����ʽΪ_______��
(2)һ��ʱ����ձ����ĸ�����˿�����滻ΪR��Ag��R��Cu����ͼB��ʾ��
�������г������ݣ��������������ɵó�Zn��Cu��Ag��R�Ļ��˳����ǿ����Ϊ______________��
����������������������R������ֺ�ɫ���壬�����Ƴ��Ľ�����___(����ĸ����)��
A. R<H B. R>Cu C. R>Zn D. R>Ag
Zn��CuSO4=== ZnSO4��Cu Zn��R��Cu��Ag C ����������̽��Zn��Cu��Ag��R���ֽ����Ļ��˳��RΪδ֪������������ͼ��֪��(1)��ͼA��ʾ�����ĸ�����˿ͬʱ�����ձ��У�������Ϊп������ͭ��Ӧ��������Ӧ�Ļ�ѧ����ʽΪ��Zn��CuSO4=ZnSO4��Cu��(2)һ��ʱ����ձ����ĸ�����˿�����滻ΪR��Ag��R��Cu����ͼB��֪���ٸ��ݡ������г������ݣ�����������...
��ϰ��ϵ�д�
�����Ŀ
������֤ʵ���ܳɹ�����(����)
|
|
|
|
A. ֤��CH4ȼ�����ɶ�����̼��ˮ | B. ��֤��˿�����������������ˮ | C. �����̬���� | D. ̽�������˶� |
A. A B. B C. C D. D
C ��������A��ʵ������У�����ʯ��ˮ����ǣ�˵������ȼ�������˶�����̼�����Dz���֤��ȼ��������ˮ����ѡ��ܳɹ���B��ʵ������У��������˿�������⣬��ʪ����˿�ܹ����⣬���Dz���֤����������Ҫˮ��������������Ϊ��˿����Ҳ����������ˮ��Ӧ�Ľ������ѡ��ܳɹ���C��ʵ������У�ʪ��ĺ�ɫʯ����ֽ����ɫ��˵����Ӧ�����˰�������һ��˵�����������Ʒ�Ӧ�����̬���ʣ���ѡ���ܹ��ɹ���D��ʵ������У�...