��Ŀ����
�Ȼ�����һ����Ҫ����Դ���ں�ˮ�д����ܷḻ��
���Ȼ�����______��Ԫ����ɣ�0.1mol �Ȼ��Ƶ�������_____g���Ȼ�������Ԫ�ص���������Ϊ___����ȷ�� 0.01����
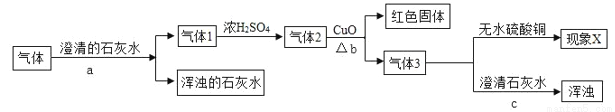
�ں�ˮɹ�εõ��Ĵ����к����������ʣ��Ƴɾ��εIJ����������£�
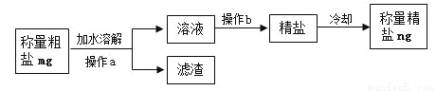
��. ��ȡ mg ������Ʒ������ʱ���ڵ�����ƽ�Ϸ�һ�ų���ֽ��Ȼ��____������_______�����������ƣ���ȡ���μӵ�����ֽ�ϣ�ֱ����ƽ��ʾ mg��
��. ���� a ������________����Һ�н�һ�������þ��Σ����� b ��������________��
��. ��֪������=(���ε������´��ε�����)x100% ʵ����С��IJ��ʼ��������£�
��� | �� 1 �� | �� 2 �� | �� 3 �� | �� 4 �� | |||
���� | 75% | 65% | 88% | 72% | |||
�Բ��ʵ����� | ƫ�� | ƫ�� | ƫ�� | ƫ�� | |||
�����Ƕ���ɲ���ƫ��ƫ�͵�ԭ�������������ȷ����_______��
A. �� 1 �飺û�����ɣ�����������ˮ�� B. �� 2 �飺����ʱ��Һ������ֽ�ı�Ե
C. �� 3 �飺����ʱ��С��ͱ������ֽ D. �� 4 �飺ʳ��δ�ܽ���Ϳ�ʼ����
�������� 25g10%��ʳ����Һ�����Ƴ� 5%��ʳ����Һ�������ˮ____g��
��ϰ��ϵ�д�
 ǧ�������������ĩ�����Ծ�����ϵ�д�
ǧ�������������ĩ�����Ծ�����ϵ�д�
�����Ŀ


 4FeO+CO2�����ֽ���̿������Fe2O3��Ͼ��ȣ��õ�211 kg�������������ԭ����ַ�Ӧ����������������Ϊ200 kg������㣺
4FeO+CO2�����ֽ���̿������Fe2O3��Ͼ��ȣ��õ�211 kg�������������ԭ����ַ�Ӧ����������������Ϊ200 kg������㣺