��Ŀ����
10����������ʱ�����һ����ɫ�г�������ζ�����壨����--H2S��������������ˮ��ˮ��Һ�����ԣ�H2S������һ�ִ�����Ⱦ����ڿ�������ȫȼ��ʱ���ɶ��������ˮ����H2S����ͨ��Ũ�����У������ķ�ӦΪ��H2S+H2SO4��Ũ���TSO2+X+2H2O��1�������������У����˽H2S��������������ɫ�г�������ζ�����壬������ˮ��
��2��д��H2S�ڿ�������ȫȼ�յĻ�ѧ����ʽ2H2S+3O2$\frac{\underline{\;��ȼ\;}}{\;}$2SO2+2H2O��
��3��H2S����ͨ��Ũ�����з�Ӧʱ���������е�x�Ļ�ѧʽΪS��
��4����β���к���H2S���壬��Ũ�����������𣿲��У���С����С��������ǣ������ɶԴ�������Ⱦ�Ķ�������
��5�������ˮ��ҺҲ���������ᣬд�������������Ʒ�Ӧ�Ļ�ѧ����ʽH2S+2NaOH=Na2S+2H2O��
���� ����Ҫͨ����ѧ�仯���ֳ����������������ʵ��������ʣ�
��ѧ��Ӧǰ��Ԫ�ص�����䣬ԭ�ӵ����ࡢ�ܸ������䣬ԭ���������䣮
��� �⣺��1�������������У�H2S��������������ɫ�г�������ζ�����壬������ˮ��
�����ɫ�г�������ζ�����壬������ˮ��
��2��H2S�ڿ�������ȫȼ�յĻ�ѧ����ʽΪ��2H2S+3O2$\frac{\underline{\;��ȼ\;}}{\;}$2SO2+2H2O��
���2H2S+3O2$\frac{\underline{\;��ȼ\;}}{\;}$2SO2+2H2O��
��3����H2S+H2SO4��Ũ���TSO2+X+2H2O��֪��
��Ӧǰ����ԭ�Ӹ�������4������ԭ�Ӹ�������4������Ӧǰ��ԭ����2������Ӧ��Ӧ����2����������X�У����X�Ļ�ѧʽ��S��
���S��
��4����β���к���H2S���壬��Ũ�������ղ��У������ǻ����ɶԴ�������Ⱦ�Ķ�������
������У������ɶԴ�������Ⱦ�Ķ�������
��5�������ˮ��ҺҲ���������ᣬ�����������Ʒ�Ӧ�Ļ�ѧ����ʽΪ��H2S+2NaOH=Na2S+2H2O��
���H2S+2NaOH=Na2S+2H2O��
���� ��ѧ��Ӧ��ѭ�����غ㶨�ɣ�����ѧ��Ӧǰ��Ԫ�ص�����䣬ԭ�ӵ����ࡢ�ܸ������䣬������д��ѧ����ʽ���ж����ʵĻ�ѧʽ���жϻ�ѧ��������������ط������Ļ�����

 100�ִ�����ĩ���ϵ�д�
100�ִ�����ĩ���ϵ�д� ��У���˿��ֿ���ϵ�д�
��У���˿��ֿ���ϵ�д�| A�� | ����200���� | B�� | ����200���� | C�� | С��200���� | D�� | ����180���� |
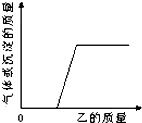 ������ļ���������μ�����ӳ������Һ����������Ӧ������������������������������ҵ�������ϵ��������ͼ��ʾ���߱�ʾ���ǣ�������
������ļ���������μ�����ӳ������Һ����������Ӧ������������������������������ҵ�������ϵ��������ͼ��ʾ���߱�ʾ���ǣ�������| ��� | �� | �� |
| �� | ͭ��п�Ļ���� | ϡ���� |
| �� | ���������ͭ�Ļ��Һ | ����������Һ |
| �� | �����ϡ����Ļ����Һ | �Ȼ�����Һ |
| �� | ��������� | ϡ���� |
| A�� | �٢� | B�� | �ڢ� | C�� | �ۢ� | D�� | ֻ�Т� |
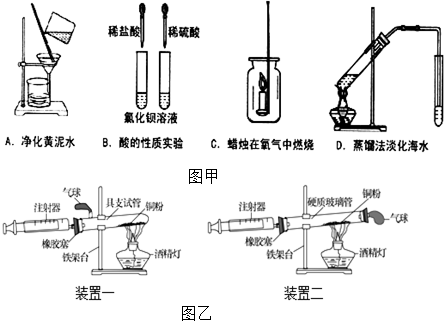
��1����Aͼ��ʾװ�þ�������ˮʱ����������������������
��2��ͨ��Bͼ��ʾ�������ʵ�飬����ʶ������Ȼ�������ʾ������ƵĻ�ѧ���ʣ������ڲ�ͬ��������ˮʱ������������ӻ�������Ӳ�ͬ��������Ҳ���ڲ��죮
��3������Cͼ��ʾ������������ȼ��ʵ��ʱ�����ü�����֤����ȼ�յIJ����ѡ��һ�ֲ����Ҫд������֤����������ƿ�ڱڳ���ˮ����֤����ˮ����
��4����Dͼ��ʾװ�ý�������ʱ��Ϊʹˮ����������Ч�����ã��ɶ�ʵ��װ������һ��Ľ��ǣ���С�Թܷ�����ˮ�У�
�̽������ʵ���ǿ�ѧ̽������Ҫ��������ͼ���Dzⶨ��������������ʵ�������װ��ͼ������ͼʾ�ش��й����⣮
��1�������±��ṩ��ʵ�����ݣ�������
| Ӳ�ʲ������п�������� | ��Ӧǰע�����п������ | ��Ӧ��ע������������� | ʵ���ÿ������������������ |
| 25mL | 15mL | 9mL | 15% |
��3����ʵ���õĽ��ƫС���������������С��21%�������ܵ�ԭ������Щ�����о�������ͭ�۵������㣻δ��װ����ȫ��ȴ�����¾Ͷ�����
| A�� | C+2CuO$\frac{\underline{\;����\;}}{\;}$ 2Cu+CO2�� | B�� | 2Mg+O2 $\frac{\underline{\;��ȼ\;}}{\;}$ 2MgO | ||
| C�� | Ba��OH��2+2HCl�TBaCl2+2H2O | D�� | NH4Cl $\frac{\underline{\;����\;}}{\;}$ NH3��+HCl�� |
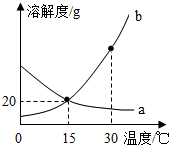 a��b �������ʵ��ܽ��������ͼ��ʾ������˵����ȷ���ǣ�������
a��b �������ʵ��ܽ��������ͼ��ʾ������˵����ȷ���ǣ�������| A�� | a��b ���ܽ�Ⱦ�Ϊ 20g | |
| B�� | 15��ʱ��a��b���ֱ�����Һ��������������һ����� | |
| C�� | ��ˮ�����¾���ʹ a �ı�����Һ��Ϊ��������Һ | |
| D�� | a���Բ�����ȴ�ȱ�����Һ�ķ����ᾧ |
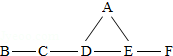 A��B��C��D��E��F�ֱ�����������ϡ���ᡢ����������Һ��̼������Һ������������Һ��������̼�е�һ�֣�B���������������֮������ͼ��ʾ�ķ�Ӧ��ϵ��ͼ�ж������������ʼ��ܷ�����Ӧ����ش�
A��B��C��D��E��F�ֱ�����������ϡ���ᡢ����������Һ��̼������Һ������������Һ��������̼�е�һ�֣�B���������������֮������ͼ��ʾ�ķ�Ӧ��ϵ��ͼ�ж������������ʼ��ܷ�����Ӧ����ش�